United States of America Regulatory Consulting Outsourcing Market Size, Share, And COVID-19 Impact Analysis, By Service Type (Regulatory Consulting & Strategy, Regulatory Writing & Publishing, Product Registration & Clinical Trial Application, and Others), By Product Category (Drugs, Medical Devices, and Biologics), By Therapeutic Area (Oncology, Immunology, Neurology, Rare Diseases, and Others) By End User (Large Pharmaceutical Companies , Small Biotechnology Companies, Medical Device Manufacturers, and Clinical Research Organisations), and United States of America Regulatory Consulting Outsourcing Market Insights, Industry Trend, Forecasts To 2035 United States of America Regulatory Consulting Outsourcing Market Insights Forecasts to 2035
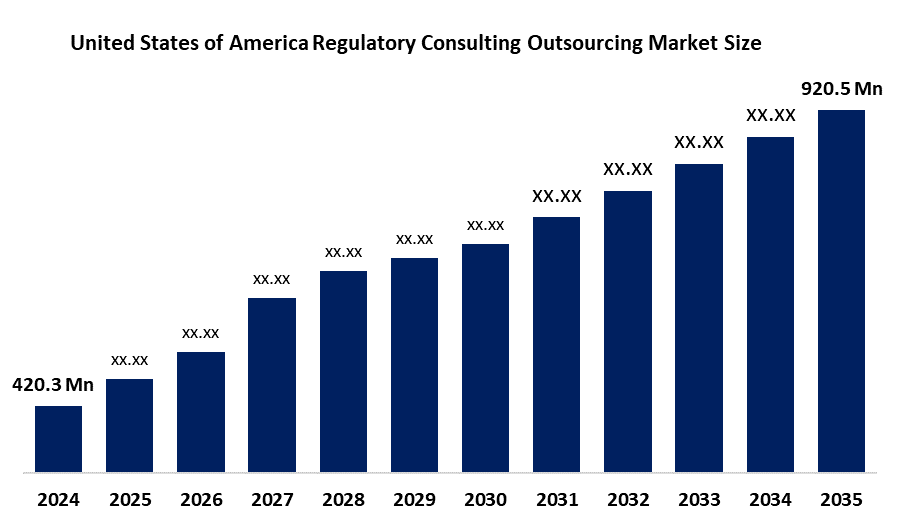
Industry: Healthcare- The United States of America Regulatory Consulting Outsourcing Market Size Was Estimated at USD 420.3 Million in 2024
- The Market Size is Expected to Grow at a CAGR of Around 8.1 % from 2025 to 2035
- The USA Regulatory Consulting Outsourcing Market Size is Expected to Reach USD 920.5 Million by 2035
Get more details on this report -
According to a research report published by Spherical Insights & Consulting, The United States of America Regulatory Consulting Outsourcing Market Size is anticipated to reach USD 920.5 Million by 2035, Growing at a CAGR of 8.1% from 2025 to 2035. The Regulatory Consulting Outsourcing market in the United States of America is driven by the rising operational costs, US FDA complexity, and faster market entry.
Market Overview
The United States regulatory consulting outsourcing market involves life sciences companies including pharmaceutical, biotechnology, and medical device firms partnering with specialized third-party experts to navigate complex FDA compliance, product registrations, and clinical trial applications. This market is experiencing robust growth, primarily driven by the increasing stringency and complexity of regulatory frameworks.
As the pipeline for advanced therapeutics (such as cell and gene therapies) and high-tech medical devices expands, many companies lack the specialized in-house expertise required to manage these novel pathways. Consequently, outsourcing provides a strategic advantage it converts high fixed internal overhead into flexible, project-based expenses while accelerating the time-to-market for critical health innovations. Technological advancements like generative AI, predictive analytics, and advanced regulatory information management systems are rapidly transforming this landscape.
Report Coverage
This research report categorizes the market for the regulatory consulting outsourcing in the United States market based on various segments and regions, and forecasts revenue growth and analyses trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the Indonesia sulphuric acid market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the Indonesia sulphuric acid market.
United States of America Regulatory Consulting Outsourcing Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 420.3 Million |
| Forecast Period: | 2024-2035 |
| Forecast Period CAGR 2024-2035 : | CAGR of 8.1% |
| 2035 Value Projection: | USD 920.5 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 210 |
| Tables, Charts & Figures: | 105 |
| Segments covered: | By Service Type, By Product Category |
| Companies covered:: | IQVIA Holdings, ICON plc, Tata Consulting Services, Rimsys intel, Parexel International, Syneos Health, Labcorp Drug Development, PPD (Part of Thermo Fisher Scientific), Fortrea, ProPharma Group, PharmaLex (part of Cencora), MCRA (Musculoskeletal Clinical Regulatory Advisers, Emergo, EAS Consulting Group, Voisin Consulting Life Sciences, Regulatory Compliance Associates, Veeva Systems, Qualio, Retina Consultants of America, and |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
The United States regulatory consulting outsourcing market is driven by factors such as the escalating complexity of FDA compliance and the sheer volume of innovative clinical trials. As pharmaceutical and medtech companies heavily invest in advanced therapeutics such as cell and gene therapies, complex biologics, and AI-enabled medical devices they face stringent, constantly evolving submission standards that are highly challenging to manage internally. To navigate these technical hurdles, organizations are increasingly partnering with specialized third-party experts, allowing them to convert high fixed overhead costs into flexible, project-based expenses. This strategic shift not only optimizes internal resources for core R&D but also significantly accelerates the time-to-market for critical health innovations and reducing the risk of costly regulatory delays. Furthermore, the widespread adoption of digital transformation tools, such as automated regulatory information management (RIM) systems and AI-driven predictive compliance models, has vastly improved the efficiency and accuracy of outsourced services.
Restraining Factors
The United States regulatory consulting outsourcing market is constrained by data security and intellectual property, as firms integrate third-party AI tools with sensitive clinical data, the potential for cyber-vulnerabilities remains a top concern, a critical talent shortage exists for experts who possess the rare dual-competency in both advanced biopharma regulations and machine learning operations and high service costs from specialized boutique firms.
Market Segmentation
The United States of America regulatory consulting market share is classified into service type, product category, therapeutic area, and end user.
- The regulatory consulting & strategy segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
Based on the service type the United States of America regulatory consulting outsourcing market is segmented into regulatory consulting & strategy, regulatory writing & publishing, product registration & clinical trial application and others. Among these the regulatory consulting & strategy segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is because the complexity of US FDA, A wrong strategy for an AI-integrated device or a therapy can lead to years of delays.
- The medical device segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
The United States of America regulatory consulting outsourcing market is segmented by product category into drugs, medical devices, and biologics. Among these, the medical device segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is because of the diversity and volume of products. Drugs and biologics can take up to 10 years to develop; medical devices have rapid 1–3-year innovation cycles. The complexity of new software as a medical device (SaMD) and the massive number of Class II devices create a constant, high-frequency need for outsourcing.
- The oncology segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
Based on the therapeutic area the United States of America regulatory consulting outsourcing market is segmented into oncology, immunology, neurology, rare diseases and others. Among these the oncology segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is because of the massive amount of global R&D capital flowing into oncology. Oncology trials are also the most complex require specialized regulatory handling which almost always necessitates external expert consultants.
- The large pharmaceutical companies segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
The United States of America regulatory consulting outsourcing market is segmented by end user into large pharmaceutical companies, small biotechnology companies, medical device manufacturers and clinical research organisations. Among these the large pharmaceutical companies segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is because the large pharmaceutical companies manage multiple products each of these requires continues regulatory maintenance. And also, Large U.S.-based pharma companies often market drugs in 50+ countries. It is nearly impossible to maintain in-house experts for every local regulatory agency (e.g. EMA in Europe, PMDA in Japan, NMPA in China). Hence the large pharmaceutical companies dominate the outsourcing market by hiring global consultancies to act as their local representatives and technical leads in these diverse territories.
Competitive Analysis:
The report offers the appropriate analysis of the key organisations/companies involved within the United States of America regulatory consulting outsourcing market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborate analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- IQVIA Holdings
- ICON plc
- Tata Consulting Services
- Rimsys intel
- Parexel International
- Syneos Health
- Labcorp Drug Development
- PPD (Part of Thermo Fisher Scientific)
- Fortrea
- ProPharma Group
- PharmaLex (part of Cencora)
- MCRA (Musculoskeletal Clinical Regulatory Advisers
- Emergo
- EAS Consulting Group
- Voisin Consulting Life Sciences
- Regulatory Compliance Associates
- Veeva Systems
- Qualio
- Retina Consultants of America
Market Segment
This study forecasts revenue at the United States, regional, and country levels from 2020 to 2035.Spherical Insights has segmented the United States of America regulatory consulting outsourcing market, based on the below-mentioned segments:
United States of America Regulatory Consulting Outsourcing Market, By Service Type
- Regulatory Consulting & Strategy
- Regulatory Writing & Publishing
- Product Registration & Clinical Trial Application
- Others
United States of America Regulatory Consulting Outsourcing Market, By Product Category
- Drugs
- Medical Devices
- Biologics
United States of America Regulatory Consulting Outsourcing Market, By Therapeutic Area Oncology
- Immunology
- Neurology
- Rare Diseases
- Others
United States of America Regulatory Consulting Outsourcing Market, By End User
- Large Pharmaceutical Companies
- Small Biotechnology Companies
- Medical Device Manufacturers and Clinical Research Organisations
Frequently Asked Questions (FAQ)
-
Q: Who are the target audiences for this market report?A: The report targets market players, investors, end-users, government authorities, consulting and research firms, venture capitalists, and value-added resellers (VARs).
-
Q: What are the key growth drivers of the market?A: Market growth is driven by escalating regulatory compliances, shortage of workforce and cyber vulnerabilities
-
Q: How is the market segmented by product category?A: The market is segmented into drugs, medical devices and biologics
Need help to buy this report?