United States of America Peripheral Vascular Devices Market Size, Share, and COVID-19 Impact Analysis, By Product Type (Peripheral Vascular Stents, Angioplasty Balloons, Plaque Modification Devices, Catheters, Inferior Vena Cava Filters, and Others), By Application (Peripheral Artery Disease, Aneurysms, Venous Diseases, and Arteriovenous Fistula), By End User (Hospitals, Ambulatory Surgical Centres, and Others), and USA Peripheral Vascular Devices Market Insights, Industry Trend, Forecasts To 2035
Industry: HealthcareUnited States of America Peripheral Vascular Devices Market Insights Forecasts to 2035
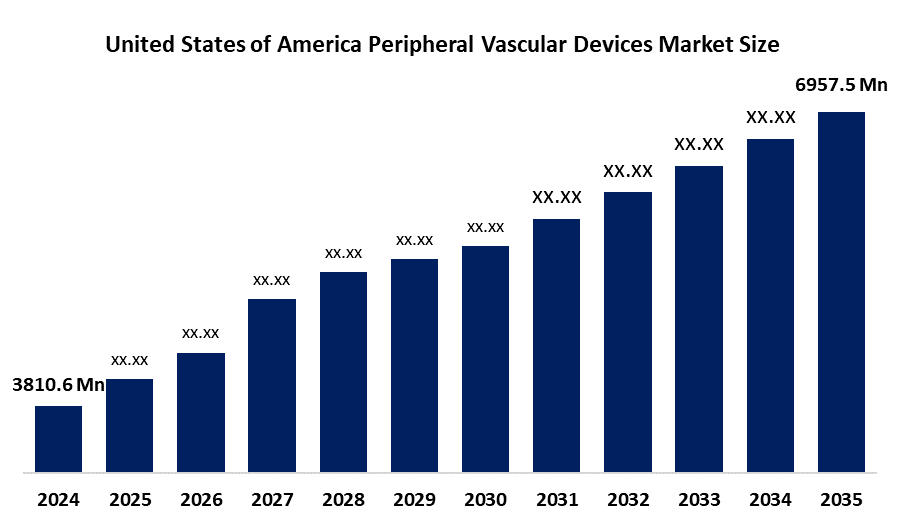
- The USA Peripheral Vascular Devices Market Size Was Estimated at USD 3,810.6 Million in 2024
- The Market Size is Expected to Grow at a CAGR of Around 5.6% from 2025 to 2035
- The USA Peripheral Vascular Devices Market Size is Expected to Reach USD 6,957.5 Million by 2035
Get more details on this report -
According to a research report published by Spherical Insights & Consulting, The USA Peripheral Vascular Devices Market Size is Anticipated to reach USD 6,957.1 million by 2035, Growing at a CAGR of 5.6% from 2025 to 2035. The peripheral vascular devices market in the USA is driven by the rising prevalence of cardiovascular conditions due to diabetes and hypertension, ageing populations, and advances in rapid diagnostic technologies.
Market Overview
The United States of America peripheral vascular device market refers to the development, manufacturing and distribution of tools designed to diagnose and treat diseases in the blood vessels outside the heart and brain. The USA peripheral vascular device market is experiencing robust expansion primarily due to the rising prevalence of peripheral arterial disease (PAD) among an ageing population and increasing rates of diabetes and obesity. As the demand for minimally invasive interventions grows, the market is shifting away from traditional open surgeries toward endovascular procedures that offer shorter recovery times and lower complication rates.
There is a rapid adoption of drug-coated balloons (DCBs) and advanced atherectomy devices over permanent metallic implants. While peripheral stents particularly drug-eluting and nitinol options remain a cornerstone of treatment for complex lesions, innovation is currently focused on reducing restenosis rates and improving long-term vessel patency. The competitive landscape is highly consolidated, with major medtech giants such as Medtronic, Boston Scientific, Abbott, and BD leading the development of next-generation devices designed to navigate complex anatomies and treat increasingly calcified lesions.
United States of America Peripheral Vascular Devices Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 3810.6 Million |
| Forecast Period: | 2024-2035 |
| Forecast Period CAGR 2024-2035 : | 5.6% |
| 2035 Value Projection: | USD 6957.5 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 240 |
| Tables, Charts & Figures: | 105 |
| Segments covered: | By Type, By Application |
| Companies covered:: | Medtronic, Boston Scientific, Becton, Dickinson and Company, Abbott Laboratories, Johnson and Johnson, Inari Medical, Strykar, Penumbra, Philips, Cook Medical, Terumo Medical Corporation, Shockwave Medical, Merit Medical Systems, Teleflex, AngioDynamics, Reflow Medical, Bolt Medical, Contego Medical, Alucent Biomedical, and Key Players. |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
The United States of America peripheral vascular devices market is driven by the rising prevalence of peripheral artery disease. This is heavily correlated with the rising incidence of diabetes and obesity. Improvements in screening and diagnosis programs by organisations like the American Heart Association are leading to earlier diagnosis of PAD. Advancements in safety and efficacy of devices have made the peripheral vascular devices safer and more effective, expanding the range of treatable patients. Cases that were previously considered untreatable or required open bypass surgery can now be managed with endovascular devices, expanding the total addressable market.
Restraining Factors
The United States of America peripheral vascular devices market is restrained by factors such as astringent regulatory landscape, as these devices are classified under class 3, high cost for development, and workforce and infrastructure challenges, as these devices require qualified personnel and complex procedures.
Market Segmentation
The United States of America peripheral vascular devices market share is classified into product type, application, and end user
- The angioplasty balloon catheter segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
Based on product type, the United States of America peripheral vascular devices market is segmented into peripheral vascular stents, angioplasty balloons, plaque modification devices, catheters, inferior vena cava filters, and others. Among these, the angioplasty balloon catheter segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is due to its lower cost, ease of use and strong familiarity of physicians.
- The peripheral artery disease segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
The United States of America peripheral vascular devices market is segmented by application into peripheral artery disease, aneurysms, venous diseases and arteriovenous fistula. Among these, the peripheral artery disease segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is due to the high prevalence of peripheral artery disease in the US, particularly among ageing, diabetic, and smoking populations. The shift toward minimally invasive endovascular therapy further strengthens this segment.
- The hospital segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
Based on the end user, the United States of America peripheral vascular devices market is segmented into hospitals, ambulatory surgical centres and others. Among these, the hospital segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is due to their capacity to manage complex peripheral interventions, including high-risk and multidevice procedures. Hospitals are also the primary adopters of advanced technologies due to their strong capital and advanced infrastructure.
Competitive Analysis:
The report offers the appropriate analysis of the key organisations/companies involved within the USA peripheral vascular devices market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborate analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Medtronic
- Boston Scientific
- Becton, Dickinson and Company
- Abbott Laboratories
- Johnson and Johnson
- Inari Medical
- Strykar
- Penumbra
- Philips
- Cook Medical
- Terumo Medical Corporation
- Shockwave Medical
- Merit Medical Systems
- Teleflex
- AngioDynamics
- Reflow Medical
- Bolt Medical
- Contego Medical
- Alucent Biomedical
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United States of America, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the USA peripheral vascular devices market based on the below-mentioned segments:
United States of America Peripheral Vascular Devices Market, By Product Type
- Peripheral Vascular Stents
- Angioplasty Balloons
- Plaque Modification Devices Catheters
- Inferior Vena Cava Filters
- Others
United States of America Peripheral Vascular Devices Market, By Application
- Peripheral Artery Disease
- Aneurysms, Venous Diseases
- Arteriovenous Fistula
United States of America Peripheral Vascular Devices Market, By End User
- Hospitals
- Ambulatory Surgical Centres
- Others
Frequently Asked Questions (FAQ)
-
Q. What is the market size of the USA peripheral vascular devices market?A. The USA peripheral vascular devices market size in 2024 was USD 3810.6 million, expected to grow at a CAGR of 5.6% to USD 6,957 million during the forecast period.
-
Q. Who are the target audiences for this market report?A: The report targets market players, investors, end-users, government authorities, consulting and research firms, venture capitalists, and value-added resellers (VARs).
-
Q. How is the market segmented by end user?A. Based on the end user, the USA peripheral vascular devices market is segmented into hospitals, ambulatory surgical centres and others.
Need help to buy this report?