Global Thoracolumbar Stabilization Devices Market Size, Share, and COVID-19 Impact Analysis, By Device Type (Posterior Stabilization Devices and Anterior Stabilization Devices), By Material (Titanium and Stainless Steel), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 - 2035
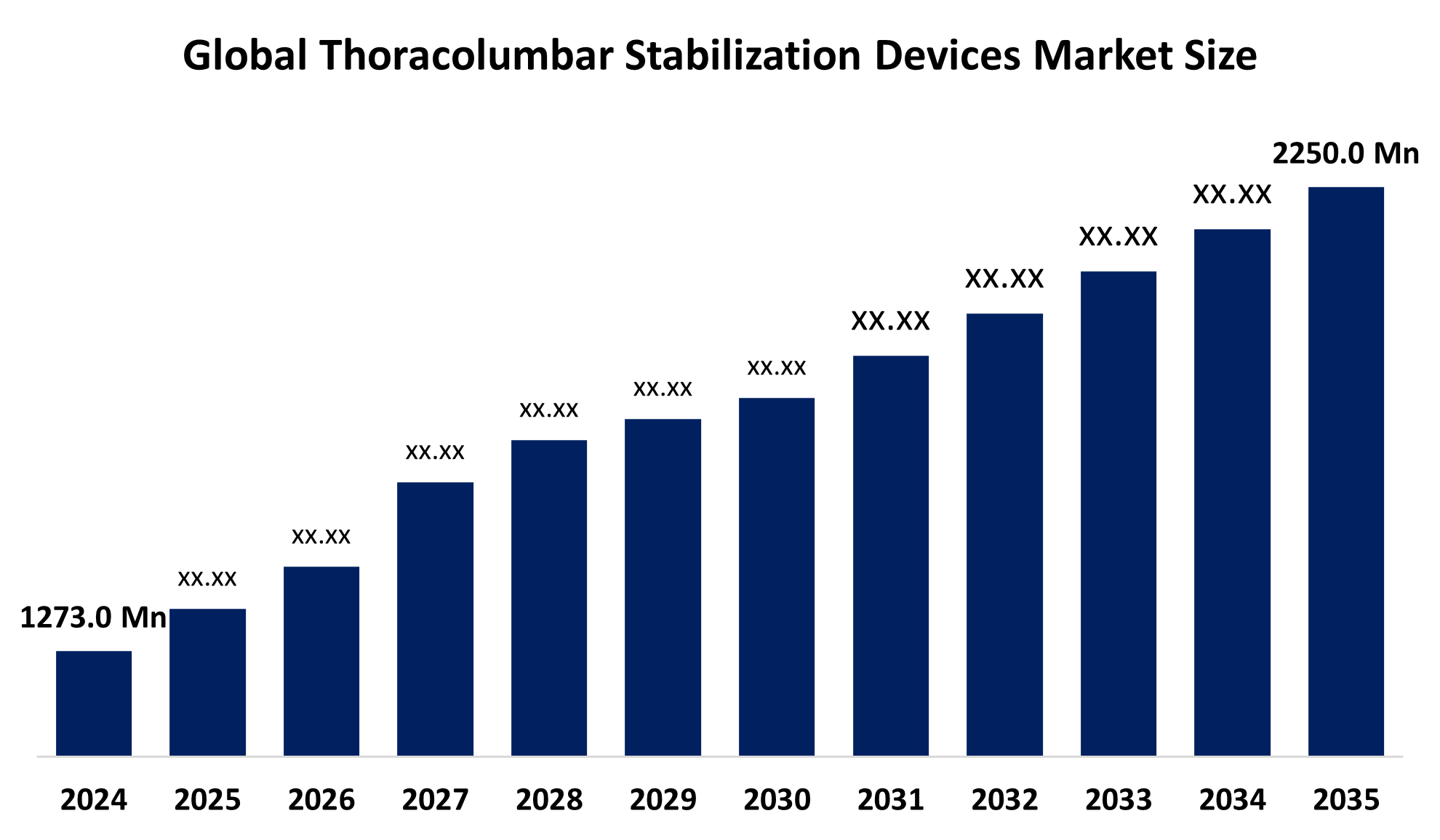
Industry: HealthcareAccording to Spherical Insights, The Global Thoracolumbar Stabilization Devices Market Size is Expected to Grow from USD 1273.0 Million in 2024 to USD 2250.0 Million by 2035, at a CAGR of 5.31% during the forecast period 2025-2035.
Key Market Trends & Opportunities
The thoracolumbar stabilization devices market has several opportunities to grow, due to the increasing technological advancements, especially in minimally invasive surgical techniques and integration of smart devices for enhanced patient efficacy and patient outcomes, awareness about spinal health, and personalized medical solutions.
- Use of bioengineered materials and enhanced imaging techniques
- Adoption of 3D printed titanium implants and robotic-assisted procedures
- Motion-preservation technologies for degenerative disc diseases and trauma
Global Thoracolumbar Stabilization Devices Market Insights Forecasts to 2035
- The Global Thoracolumbar Stabilization Devices Market Size Was Estimated at USD 1273.0 Million in 2024
- The Market Size is Expected to Grow at a CAGR of around 5.31% from 2025 to 2035
- The Worldwide Thoracolumbar Stabilization Devices Market Size is Expected to Reach USD 2250.0 Million by 2035
Get more details on this report -
Major Players
Medtronic plc, DePuy Synthes, Stryker Corporation, Globus Medical, Zimmer Biomet Holdings, NuVasive, Inc., B. Braun Melsungen AG, Orthofix Medical Inc., Alphatec Spine, Inc., and Aesculap Implant Systems
Market Overview of Thoracolumbar Stabilization Devices
The global industry for thoracolumbar stabilization devices encompasses medical implants used for stabilizing the thoracic and lumbar spine (middle and lower back) following injury, degenerative disease, or deformity. Thoracolumbar stabilization devices are surgical implants like pedicle screws, rods, and cages used for stabilizing, aligning, and supporting the spine between the thoracic and lumbar regions (T10-L2). The growing prevalence of spinal disorders, traumatic injuries, and age-related degenerative conditions is driving the significant growth in the thoracolumbar stabilization devices market. The increasing world’s ageing population, which increases the risk of degenerative disc disease, scoliosis, spinal stenosis, and vertebral compression fractures, is another factor driving up demand for these devices. Furthermore, advancements in minimally invasive surgical techniques like robotic-assisted systems and bioengineered materials are enhancing surgical precision and reducing postoperative recovery times.
Innovation and market expansion are anticipated as a result of major players' growing R&D expenditures and expanding partnerships. For instance, in December 2025, Companion Spine, the French-American specialist in surgeon-innovated spine implant surgery, announced that the U.S. Food and Drug Administration had approved the DIAM Spinal Stabilization System (P240043).
Report Coverage
This research report categorizes the thoracolumbar stabilization devices market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the thoracolumbar stabilization devices market. Recent market developments and competitive strategies such as expansion, type launch, development, partnership, merger, and acquisition have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the thoracolumbar stabilization devices market.
Global Thoracolumbar Stabilization Devices Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 1273.0 Million |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | 5.31% |
| 2035 Value Projection: | USD 2250.0 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 225 |
| Tables, Charts & Figures: | 102 |
| Segments covered: | By Device Type, By Material |
| Companies covered:: | Medtronic plc, DePuy Synthes, Stryker Corporation, Globus Medical, Zimmer Biomet Holdings, NuVasive, Inc., B. Braun Melsungen AG, Orthofix Medical Inc., Alphatec Spine, Inc., Aesculap Implant Systems and Other Key Players |
| Pitfalls & Challenges: | and COVID-19 Impact Analysis |
Get more details on this report -
Driving Factors
Prevalence of spinal disorders
Spinal disorders are a leading cause of global disability, with low back pain (LBP) affecting 619 million people in 2020, expected to rise to 843 million by 2050. In 2021, nearly 14.5 million people lived with spinal cord injuries (SCI), due to ageing populations and sedentary lifestyles. Thus, an increased prevalence of spinal disorders, along with an ageing population and increasing obesity rates, is expected to drive the thoracolumbar stabilization devices market demand.
Technological advancement
This includes robotic-assisted systems and bioengineered materials that enhance surgical precision and reduce postoperative recovery times. Innovations like smart, sensor-enabled implants for real-time monitoring and advanced biomaterials improving fusion rates, reducing operating times, blood loss, and complication rates.
Restraining Factors
Post-surgical complications and Implant failures
Complex biomechanics, increased complication rates, and the upsurging demand for personalized approaches are challenging the market. However, for early mobilization and structural support, the devices carry disadvantages like hardware failure and adjacent segment degeneration.
Market Segment Insights
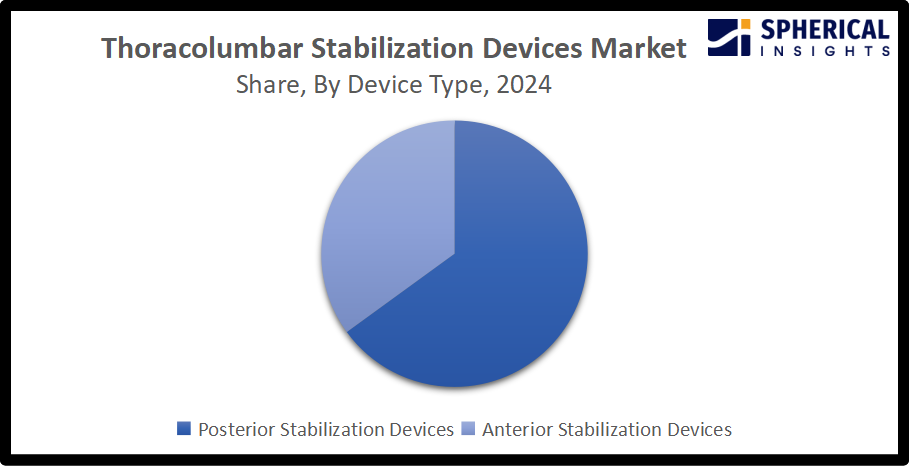
By Device Type: Posterior Stabilization Devices (Largest) vs Anterior Stabilization Devices (Emerging)
The posterior stabilization devices segment is dominating the market with a share of about 65.0% in 2024, owing to the procedural simplicity and anatomical accessibility. For instance, the S4 Element Spine System is a low-profile and innovative pedicle fixation system designed for addressing complex posterior pathologies. An increasing adoption of the devices in minimally invasive procedures is expanding their usage across outpatient settings, thereby propelling the segmental market growth. On the contrary, anterior stabilization devices include specialized plates, screws, and structural cages used to support the spine’s anterior column following fractures, tumors, or decompression.
Get more details on this report -
By Material: Titanium (Largest) vs Stainless Steel (Emerging)
Titanium is identified as a leading segment capturing around 75.0% share in 2024, driven by the material’s extensive strength-to-weight ratio, biocompatibility, and corrosion resistance. Titanium is a standard material for anterior column reconstruction (vertebral body replacement) and posterior fixation (rods and screws). While stainless steel is increasingly adopted among healthcare providers due to its high rigidity, strength, and corrosion resistance.
Regional Segment Analysis of the Thoracolumbar Stabilization Devices Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
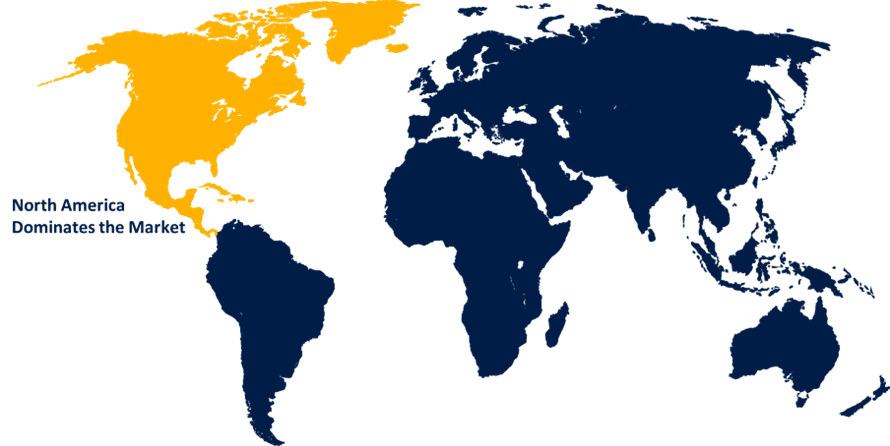
North America is anticipated to hold the largest share of the thoracolumbar stabilization devices market over the predicted timeframe.
Get more details on this report -
North America is anticipated to hold the largest share of about 45.0% in the thoracolumbar stabilization devices market over the predicted timeframe. The market ecosystem in North America is strong, due to increased spinal disorder prevalence, healthcare infrastructure, and ongoing product innovation. The demand for thoracolumbar stabilization devices has been driven by the region's aging population and surge in, traumatic and sports-related injuries. The United States is leading the North America thoracolumbar stabilization devices market, owing to the increased need for spinal fusion surgeries.
Asia Pacific is expected to grow at a rapid CAGR of about 8.25% in the thoracolumbar stabilization devices market during the forecast period. The Asia Pacific area has a thriving market for thoracolumbar stabilization devices due to its ageing population, increasing spinal disorder cases, and adoption of MIS techniques. Increased healthcare spending by the governments has also fueled the expansion of the thoracolumbar stabilization devices market. China is the dominant country, capturing the largest share, due to its massive patient base, increased surgical volume, and expanding healthcare infrastructure.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the thoracolumbar stabilization devices market, along with a comparative evaluation primarily based on their type of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes type development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Medtronic plc
- DePuy Synthes
- Stryker Corporation
- Globus Medical
- Zimmer Biomet Holdings
- NuVasive, Inc.
- B. Braun Melsungen AG
- Orthofix Medical Inc.
- Alphatec Spine, Inc.
- Aesculap Implant Systems
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting And Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Industry Development
- In July 2025, Companion Spine LLC, the French-American specialist in spine pain-management surgery, announced that it had entered into a definitive agreement to acquire the Coflex Interlaminar Stabilization device and CoFix Posterior MIS Fusion System implants, Paradigm Spine GmbH, its subsidiaries and all Paradigm Spine-related business activity globally from Xtant Medical Holdings, Inc.
- In March 2025, Spinal Stabilization Technologies, LLC (SST) announced a significant milestone in its mission to revolutionize spinal care: the enrollment of the first U.S. patient in its Investigational Device Exemption (IDE) clinical trial for the PerQdisc Nucleus Replacement Device (NRD).
- In September 2024, Xtant Medical Holdings, Inc., a leading global medical technology company focused on the treatment of spinal disorders, announced the launch of the Cortera Posterior Fixation System, a comprehensive solution designed to streamline thoracolumbar fixation surgeries.
- In August 2024, Globus Medical, Inc., a leading musculoskeletal solutions company, announced the commercial launch of the ADIRA XLIF Plate System, the Company’s first product launch compatible across its expanded lateral interbody portfolio.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the thoracolumbar stabilization devices market based on the below-mentioned segments:
Global Thoracolumbar Stabilization Devices Market, By Device Type
- Posterior Stabilization Devices
- Anterior Stabilization Devices
Global Thoracolumbar Stabilization Devices Market, By Material
- Titanium
- Stainless Steel
Global Thoracolumbar Stabilization Devices Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
-
1.What is the market size of the thoracolumbar stabilization devices market?The global thoracolumbar stabilization devices market size is expected to grow from USD 1273.0 Million in 2024 to USD 2250.0 Million by 2035, at a CAGR of 5.31% during the forecast period 2025-2035.
-
2.Which region holds the largest share of the thoracolumbar stabilization devices market?North America is anticipated to hold the largest share of the thoracolumbar stabilization devices market over the predicted timeframe.
-
3.What is the forecasted CAGR of the Global thoracolumbar stabilization devices market from 2024 to 2035?The market is expected to grow at a CAGR of around 5.31% during the period 2024–2035.
-
4.Who are the top companies that are involved in the Global thoracolumbar stabilization devices Market?Key players include Medtronic plc, DePuy Synthes, Stryker Corporation, Globus Medical, Zimmer Biomet Holdings, NuVasive, Inc., B. Braun Melsungen AG, Orthofix Medical Inc., Alphatec Spine, Inc., and Aesculap Implant Systems.
-
5.What are the main drivers in the thoracolumbar stabilization devices market?An increasing prevalence of spinal disorders, as well as technological advancements like robotic-assisted systems and bioengineered materials, are major market growth drivers of the thoracolumbar stabilization devices market.
-
6.What challenges are limiting the adoption of thoracolumbar stabilization devices treatment?Post-surgical complications and implant failure, along with complex biomechanics, increased complication rates, and the upsurging demand for personalized approaches, remain key restraints in the thoracolumbar stabilization devices market.
-
7.What are the key trends in the thoracolumbar stabilization devices market?The use of bioengineered materials, adoption of 3D printed titanium implants & robotics, and motion preservation technologies are major key trends in the thoracolumbar stabilization devices market.
Need help to buy this report?