Global Pediatric Influenza Treatment Market Size, Share, and COVID-19 Impact Analysis, By Treatment Type (Neuraminidase Inhibitors, Adamantanes, Amantadine & Rimantadine, and Oseltamivir & Zanamivir), By End User (Hospitals and Clinics), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 - 2035
Industry: HealthcareGlobal Pediatric Influenza Treatment Market Size Insights Forecasts to 2035
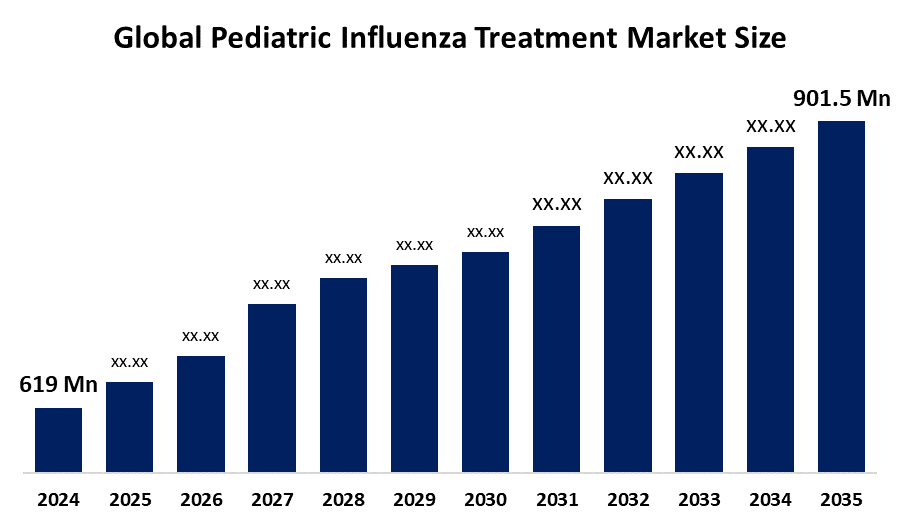
- The Global Pediatric Influenza Treatment Market Size Was Estimated at USD 619.0 Million in 2024
- The Market Size is Expected to Grow at a CAGR of around 3.48% from 2025 to 2035
- The Worldwide Pediatric Influenza Treatment Market Size is Expected to Reach USD 901.5 Million by 2035
- Asia Pacific is expected to Grow the fastest during the forecast period.
Get more details on this report -
According to a Research Report Published by Spherical Insights and Consulting, The Global Pediatric Influenza Treatment Market Size was worth around USD 619.0 Million in 2024 and is predicted to Grow to around USD 901.5 Million by 2035 with a compound annual growth rate (CAGR) of 3.48% from 2025 and 2035. The market for pediatric influenza treatment has a number of opportunities to grow due to the increasing influenza activity and demand for more effective novel therapeutic drugs. There is a shift towards faster-acting, single-dose antivirals and novel therapies targeting viral replication. The development of these drug therapeutics for enhanced efficacy and duration of protection is anticipated to propel growth in the pediatric influenza treatment market.
Market Overview
The global industry for pediatric influenza encompasses antiviral drugs (e.g., oseltamivir, zanamivir, baloxavir) and supportive care products designed to reduce symptom duration and prevent complications in children. It was estimated that the incidence of influenza in children under the age of 5 is 90 million cases per year, estimating 19/1000 per year incidence in the U.S. Children under the age of five years old are hospitalized at a rate of 1000 per 100,000 person-years due to influenza and related disorders, making them the largest group of patients. Due to high infection rates, there is a growing need for earlier, increased antiviral use, especially for high-risk children. Pediatric influenza treatment emphasizes the easing symptoms through rest, hydration, and fever reduction (acetaminophen or ibuprofen). While antiviral medications like oseltamivir are recommended for severe illness, ideally within 48 hours of symptom onset to reduce duration and severity.
Innovation and market expansion are anticipated as a result of major players' growing R&D expenditures and expanding partnerships. For instance, in February 2023, WILEY Online Library published a research article on the knockout of Noxa with CRISPR/Cas9, increasing host resistance to influenza virus infection.
Report Coverage
This research report categorizes the pediatric influenza treatment market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the pediatric influenza treatment market. Recent market developments and competitive strategies such as expansion, type launch, development, partnership, merger, and acquisition have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the pediatric influenza treatment market.
Global Pediatric Influenza Treatment Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024 : | USD 619.0 Million |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | 3.48% |
| 2035 Value Projection: | USD 901.5 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 283 |
| Tables, Charts & Figures: | 109 |
| Segments covered: | By Treatment Type, By End User and COVID-19 Impact Analysis |
| Companies covered:: | GlaxoSmithKline Plc, NATCO Pharma Limited, NESHER PHARMS, F. Hoffmann-La Roche Ltd, Atabay Kimya Sanayi ve Ticaret A.S., Cipla, Hetero Pharma, Teva Pharmaceutical pvt. Ltd, Olainfarm JSC, and Others |
| Pitfalls & Challenges: | COVID-19 Empact, Challenges, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
The market for pediatric influenza treatment is driven by the increasing severity of pediatric flu cases. Implementing early intervention strategies by starting antiviral treatment within 48 hours of symptoms starting is highly effective in reducing mortality and other complications. Novel rapid viral diagnostic tests, including molecular diagnostics tests, for selecting optimal anti-infective therapy also contributing to promoting the pediatric influenza treatment market. Additionally, government initiatives and vaccination programs aid in reducing the chances of causing influenza in children, thereby propelling the market growth. With the growing healthcare infrastructure, innovations in antiviral therapies are anticipated to escalate market growth. For instance, the Pharmaceutical Society of Australia (PSA) has welcomed the Cook Government’s $4.78 million investment to secure 130,000 doses of the FluMist intranasal influenza vaccine for children aged two to under 12 years ahead of the 2026 flu season.
Restraining Factors
The pediatric influenza treatment market is restricted by factors like increased cost of branded medications, inefficient usage of low-cost non-prescribed drugs, and limited accessibility. Further, the limited availability of treatment options for pediatric influenza is challenging the market.
Market Segmentation
The pediatric influenza treatment market share is classified into treatment type and end user.
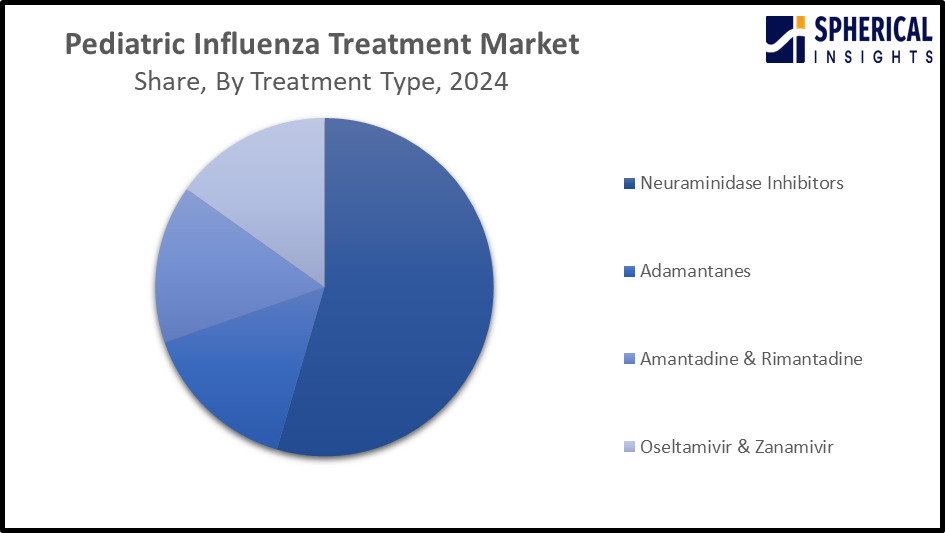
- The neuraminidase inhibitors segment dominated the market with the largest share of around 53.9% in 2024 and is projected to grow at a substantial CAGR during the forecast period.
Based on the treatment type, the pediatric influenza treatment market is divided into neuraminidase inhibitors, adamantanes, amantadine & rimantadine, and oseltamivir & zanamivir. Among these, the neuraminidase inhibitors segment dominated the market with the largest share of around 53.9% in 2024 and is projected to grow at a substantial CAGR during the forecast period. Neuraminidase inhibitors are recommended for children hospitalized with influenza-related respiratory infections. Oseltamivir is the first choice of treatment in most cases. The widespread application of neuraminidase inhibitors for reducing the duration and severity of influenza symptoms is contributing to driving segmental market growth.
Get more details on this report -
- The hospitals segment accounted for the largest share of about 63.5% in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the end user, the pediatric influenza treatment market is divided into hospitals and clinics. Among these, the hospitals segment accounted for the largest share of about 63.5% in 2024 and is anticipated to grow at a significant CAGR during the forecast period. It was estimated that on average, influenza accounts for nearly 1 million hospitalizations worldwide in children aged younger than 5 years. Advancements in diagnostic capabilities for timely identification and treatment, which improve patient outcomes, are enhancing the market growth in the hospitals segment.
Regional Segment Analysis of the Pediatric Influenza Treatment Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the Pediatric Influenza Treatment market over the predicted timeframe.
North America is anticipated to hold the largest share of about 34.0% in the pediatric influenza treatment market over the predicted timeframe. The market is driven by the development of innovative treatments with safe, effective, and age-appropriate flu medication, as well as an increased prevalence of influenza among pediatric populations. Furthermore, an increasing awareness among parents and healthcare providers about the significance of flu vaccination and early intervention in vulnerable groups in children plays an important role in enhancing the market growth of pediatric influenza treatment. The United States is dominating the pediatric influenza treatment market within the North America region, due to the ongoing awareness and adoption of advanced drug delivery systems.
Asia Pacific is expected to grow at a rapid CAGR of nearly 22.0% in the pediatric influenza treatment market during the forecast period. The Asia Pacific area has a thriving market for pediatric influenza treatment due to its increasing need for healthcare infrastructure and advancements in antiviral drug development. Due to the expanding usage of influenza vaccines in the Asia Pacific region and an increasing improvement in current vaccine technologies, the market for pediatric influenza treatment is anticipated to be propelled. China is the major contributor to the Asia Pacific pediatric influenza treatment market, owing to its large population base, vaccination programs, and ongoing clinical research studies on drug medication.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the pediatric influenza treatment market, along with a comparative evaluation primarily based on their type of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes type development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- GlaxoSmithKline Plc
- NATCO Pharma Limited
- NESHER PHARMS
- F. Hoffmann-La Roche Ltd
- Atabay Kimya Sanayi ve Ticaret A.S.
- Cipla
- Hetero Pharma
- Teva Pharmaceutical pvt. Ltd
- Olainfarm JSC
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting And Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In July 2025, ArkBio's Ziresovir Included into Pediatric RSV Drug Priority List by WHO. In the newly released "Paediatric drug optimization for respiratory syncytial virus (PADO-RSV) meeting report" by Global Accelerator for Paediatric formulations (GAP-f), the World Health Organization (WHO) host network, ziresovir (10mg) has been included in the PADO-RSV priority list.
- In April 2024, TaiGen Biotechnology Company, Ltd. announced the successful completion of the phase III study of the influenza antiviral TG-1000 by its partner, Joincare Pharmaceutical.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the Pediatric Influenza Treatment market based on the below-mentioned segments:
Global Pediatric Influenza Treatment Market, By Treatment Type
- Neuraminidase Inhibitors
- Adamantanes
- Amantadine & Rimantadine
- Oseltamivir & Zanamivir
Global Pediatric Influenza Treatment Market, By End User
- Hospitals
- Clinics
Global Pediatric Influenza Treatment Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
-
1. What is the market size of the pediatric influenza treatment market?The global pediatric influenza treatment market size is expected to grow from USD 619.0 Million in 2024 to USD 901.5 Million by 2035, at a CAGR of 3.48% during the forecast period 2025-2035.
-
2. Which region holds the largest share of the pediatric influenza treatment market?North America is anticipated to hold the largest share of the pediatric influenza treatment market over the predicted timeframe.
-
3. What is the forecasted CAGR of the Global Pediatric Influenza Treatment Market from 2024 to 2035?The market is expected to grow at a CAGR of around 3.48% during the period 2024–2035.
-
4. Who are the top companies that are involved in the Global Pediatric Influenza Treatment Market?Key players include GlaxoSmithKline Plc, NATCO Pharma Limited, NESHER PHARMS, F. Hoffmann-La Roche Ltd, Atabay Kimya Sanayi ve Ticaret A.S., Cipla, Hetero Pharma, Teva Pharmaceutical pvt. Ltd, and Olainfarm JSC.
-
5. What are the main drivers in the pediatric influenza treatment market?An increasing severity of pediatric flu cases, implementing early intervention strategies, novel rapid viral diagnostic tests, government initiatives and vaccination programs, and innovation are major market growth drivers of the pediatric influenza treatment market.
-
6. What challenges are limiting the adoption of pediatric influenza treatment treatment?Factors like increased cost of branded medications, inefficient usage of low-cost non-prescribed drugs, and limited accessibility remain key restraints in the pediatric influenza treatment market.
-
7. What are the key trends in the pediatric influenza treatment market?The demand for more effective novel therapeutic drugs, shift towards rapid point-of-care diagnostics are key trends in the pediatric influenza treatment market.
Need help to buy this report?