North America Hepatitis Therapeutics Market Size, Share, and COVID-19 Impact Analysis, By Disease Type (Hepatitis A, Hepatitis B, Hepatitis C, and Others), By Application (Hospital Pharmacy, Retail Pharmacy, and Others), and North America Hepatitis Therapeutics Market Insights, Industry Trend, Forecasts to 2035
Industry: HealthcareNorth America Hepatitis Therapeutics Market Insights Forecasts to 2035
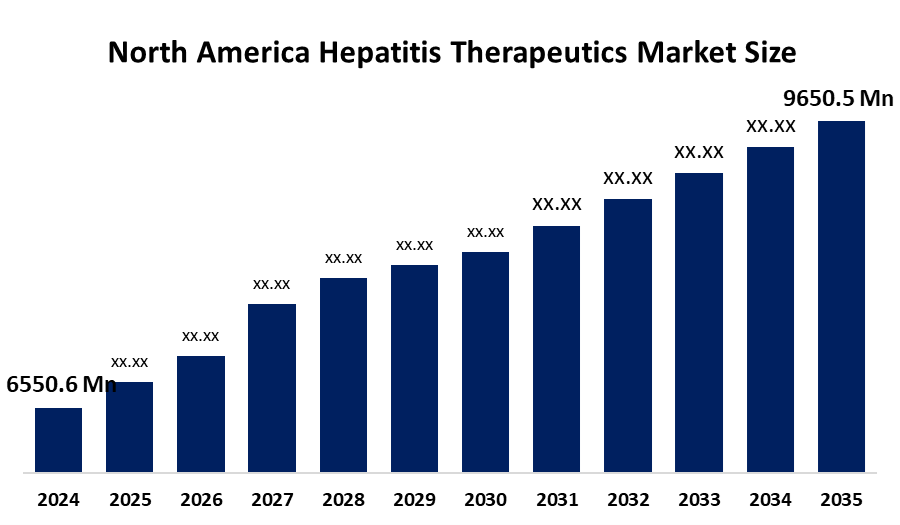
- The North America Hepatitis Therapeutics Market Size Was Estimated at USD 6550.6 Million in 2024
- The Market Size is Expected to Grow at a CAGR of Around 3.59% from 2025 to 2035
- The North America Hepatitis Therapeutics Market Size is Expected to Reach USD 9650.5 Million by 2035
Get more details on this report -
According to a research report published by Spherical Insights & Consulting, the North America Hepatitis Therapeutics market size is anticipated to reach USD 9650.5 million by 2035, growing at a CAGR of 3.59% from 2025 to 2035. The hepatitis therapeutics market in North America is driven by increased innovation in drug discovery with the adoption of AI, development of novel antiviral compounds and favourable government policies to inhibit the disease.
Market Overview
The North America hepatitis therapeutics market refers to the pharmaceutical and diagnostic ecosystem dedicated to treating inflammation of the liver caused by viral infections (Hepatitis A, B, C, D, and E), autoimmune conditions, or substance abuse. the market is defined by a high diagnosis rate and the rapid adoption of Direct-Acting Antivirals (DAAs). While the Hepatitis C (HCV) segment has matured due to cure rates exceeding 95%, the focus in 2026 has shifted toward the Hepatitis B (HBV) and Delta (HDV) sectors, where chronic infection remains a significant clinical hurdle.
Innovation is the primary growth factor of this market, with Artificial Intelligence (AI) now deeply integrated into drug discovery to predict treatment outcomes and identify novel antiviral compounds. The industry is witnessing a therapeutic resurgence with the rise of RNA interference (RNAi) and Antisense Oligonucleotides (ASOs), such as bepirovirsen, which aim to achieve a functional cure for HBV by silencing viral gene expression. Furthermore, in vivo gene-editing trials (using CRISPR-based technologies) are currently exploring ways to permanently excise viral DNA from the liver. To improve patient compliance, the market is also pivoting toward long-acting injectables that reduce the daily pill burden to a single dose every few months.
Public health policies in the United States and Canada have become increasingly aggressive. A cornerstone of this effort is the national Hepatitis C Elimination initiative, supported by the Cure Hepatitis C Act, which leverages subscription-style payment models to provide universal access to treatments. Federal agencies like the CDC and HHS have extended their strategic plans through 2030, emphasizing expanded screening in primary care and decentralizing treatment through tele-hepatology. These initiatives, combined with FDA Breakthrough Therapy designations, ensure that the pipeline for life-saving therapeutics remains robust and financially accessible.
Report Coverage
This research report categorizes the North America hepatitis therapeutics market based on various segments and regions, and forecasts revenue growth and analyses trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the North America hepatitis therapeutics market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the Indonesia sulphuric acid market.
North America Hepatitis Therapeutics Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 6550.6 Million |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | 3.59% |
| 2035 Value Projection: | USD 9650.5 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 209 |
| Tables, Charts & Figures: | 123 |
| Segments covered: | By Disease Type, By Application |
| Companies covered:: | Gilead Sciences, GSK, AbbVie, Bristol Myers Squibb, Johnson and Johnson, Merck, Arbutus Biopharma, Alnylam Pharmaceuticals, Assembly Biosciences, Excision Biotherapeutics, Others, |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
The North America hepatitis therapeutics is driven by factors such as the democratization of specialized care, where the traditional bottleneck of hepatologist-only prescriptions has been dismantled. Through AI-enhanced clinical decision support systems, primary care providers can now manage complex antiviral regimens with high confidence, effectively expanding the prescriber base. Simultaneously, the market is responding to innovative reimbursement models such as subscription-based state contracts that allow for unlimited drug access for a flat fee which has decoupled profit from pill volume and incentivized mass treatment.
Furthermore, there is a significant intersectional clinical push by integrating hepatitis screening and therapy directly into opioid recovery and harm-reduction centres, providers are capturing a high-risk demographic that was previously lost to follow-up. This is strengthened by the pharmacogenomic revolution, where rapid genetic testing allows for the selection of the most effective Direct-Acting Antiviral (DAA) based on an individual’s viral resistance profile, virtually guaranteeing a cure on the first attempt.
Restraining Factors
The North America hepatitis therapeutics market is constrained by various factors such as the high cost of development of novel gene-editing and RNAi therapies also extends time-to-market for potential functional cures, high out-of-pocket costs and restrictive insurance prior-authorization protocols continue to create a barrier, preventing thousands of diagnosed patients from initiating therapy.
Market Segmentation
The North America hepatitis therapeutics market share is classified into disease type and distribution channel.
- The hepatitis C segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
The North America hepatitis therapeutics market is segmented by disease type into hepatitis A, hepatitis B, hepatitis C, and others Among these, the hepatitis C segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is due to the widespread use of high-value Direct-Acting Antiviral (DAA) combination therapies, which command premium pricing due to their curative nature. HCV generates higher immediate revenue through intensive, short-term treatment regimens. The segment's lead is further bolstered by aggressive federal test-and-treat mandates and the clearing of a massive backlog of patients who were previously ineligible for older, less effective therapies.
- The hospital pharmacy segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period.
The North America hepatitis therapeutics market is segmented by distribution channel into hospital pharmacy, retail pharmacy, and others. Among these, the hospital pharmacy segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. Because hospitals serve as the central hubs for the initial diagnosis, staging of liver fibrosis, and the management of advanced complications like cirrhosis or hepatocellular carcinoma. Given the high cost and specialized storage requirements of advanced antiviral drugs, patients and payers often prioritize hospital-based pharmacies to ensure integrated care and supervised administration.
Competitive Analysis:
The report offers the appropriate analysis of the key organisations/companies involved within the North America hepatitis therapeutics market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborate analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Gilead Sciences
- GSK
- AbbVie
- Bristol Myers Squibb
- Johnson and Johnson
- Merck
- Arbutus Biopharma
- Alnylam Pharmaceuticals
- Assembly Biosciences
- Excision Biotherapeutics
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Developments:
- In December 2025, Sanofi announced that it has entered into an agreement to acquire Dynavax Technologies Corporation (Dynavax), a publicly traded vaccines company with a marketed adult hepatitis B vaccine (HEPLISAV-B) and differentiated shingles vaccine candidate. The acquisition augments Sanofi’s presence in adult immunization by bringing together Dynavax’s vaccines with Sanofi’s global scale, development capabilities and commercial reach.
Market Segment
This study forecasts revenue at the North America, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the North America hepatitis therapeutics market based on the below-mentioned segments:
North America Hepatitis Therapeutics Market, By Disease Type
- Hepatitis A
- Hepatitis B
- Hepatitis C
- Others
North America Hepatitis Therapeutics Market, By Application
- Hospital Pharmacy
- Retail Pharmacy
- Others
Frequently Asked Questions (FAQ)
-
Q1: What is the current and projected market size of the North America hepatitis therapeutics market?A: The market was estimated at USD 6550.6 million in 2024 and is projected to reach USD 9650.5 million by 2035. This growth represents a Compound Annual Growth Rate (CAGR) of 3.59% during the forecast period from 2025 to 2035.
-
Q2: Which disease type is the fastest growing segment in the North America hepatitis therapeutics market?A: The Hepatitis B (HBV) segment accounted for the fastest growing revenue market share in 2024. Hepatitis B (HBV) holds due to the chronic, lifelong need for viral suppression
-
Q3: Which distribution channel is the leading segment in this market?A: The hospital pharmacy segment is the dominant application distribution channel. This is due to hospitals acting as primary hubs for diagnosis, liver fibrosis staging, and managing advanced complications, alongside their ability to handle the specialized storage and high costs of antiviral drugs.
-
Q4: What are the primary factors driving growth in the North America hepatitis therapeutics industry?A: Key drivers include the integration of Artificial Intelligence (AI) in drug discovery, the development of novel compounds like RNA interference (RNAi) and Antisense Oligonucleotides (ASOs), and aggressive government initiatives like the Cure Hepatitis C Act and subscription-style reimbursement models.
-
Q5: What are the main restraining factors affecting the market?A: Market growth is constrained by the high cost of developing gene-editing and RNAi therapies, which extends time-to-market. Additionally, high out-of-pocket costs and restrictive insurance prior-authorization protocols create barriers that prevent many diagnosed patients from starting therapy.
Need help to buy this report?