North America Antithrombin Market Size, Share, and COVID-19 Impact Analysis, By Application (Therapeutics, Diagnostics, Research, and Others), By Source (Human Derived, Recombinant Goat Milk, and Others), By Thrombin Type (Anticoagulant, Antiplatelets, Thrombolytic Drugs, and Others), and North America antithrombin Market Insights, Industry Trend, Forecasts to 2035
Industry: HealthcareNorth America Antithrombin Market Insights Forecasts to 2035
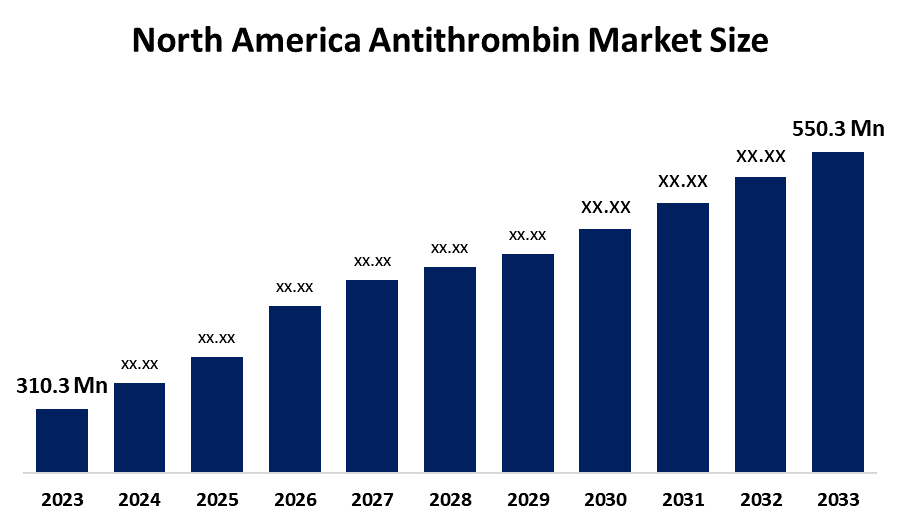
- The North America antithrombin Market Size Was Estimated at USD 310.3 Million in 2024
- The Market Size is Expected to Grow at a CAGR of Around 5.35% from 2025 to 2035
- The North America antithrombin Market Size is Expected to Reach USD 550.3 Million by 2035
Get more details on this report -
According to a research report published by Spherical Insights & Consulting, The North America Antithrombin Market Size Is Anticipated To Reach USD 550.3 Million By 2035, Growing At A CAGR Of 5.35% From 2025 To 2035. The antithrombin market in North America is driven by rising geriatric population susceptible to thromboembolic events, the increasing prevalence of hereditary ATIII deficiencies, massive investments in development of precision medicine and sophisticated healthcare infrastructure
Market Overview
The North America Antithrombin Market Size refers to Antithrombin III (ATIII) a vital plasma protein that functions as a natural anticoagulant by neutralizing thrombin and other clotting factors. According to data from the Centre for Disease Control and Prevention (CDC), venous thromboembolism (VTE) impacts up to 900,000 Americans annually, while the National Institutes of Health (NIH) estimates that hereditary deficiency affects roughly 1 in 2,000 to 3,000 individuals. A primary driving factor is the high volume of complex cardiovascular surgeries; for instance, the CDC reports nearly 395,000 coronary artery bypass grafts performed each year, this market is currently transitioning from traditional plasma-derived extraction toward recombinant innovations, such as transgenic platforms that produce the protein in animal milk, ensuring a more scalable and pathogen-free supply.
Growth is significantly bolstered by government incentives, including the FDA’s Orphan Drug Designation and Research & Development tax credits, which mitigate the high costs associated with developing therapies for rare haematological conditions. Furthermore, federal funding through the NIH and clinical grants provides a safety net for biotechnology firms exploring expanded indications. The primary driving factors include a rising geriatric population susceptible to thromboembolic events, the increasing prevalence of hereditary ATIII deficiencies, and the expanding use of antithrombin in complex procedures like cardiopulmonary bypass surgery. As the region prioritizes precision medicine, the integration of advanced diagnostic screening is further accelerating the demand for targeted anticoagulant therapies across the United States and Canada.
Report Coverage
This research report categorizes the market for the North America antithrombin market based on various segments and regions, and forecasts revenue growth and analyses trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the North America antithrombin market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the North America antithrombin market.
North America Antithrombin Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 310.3 Million |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | CAGR Of 5.35% |
| 2035 Value Projection: | USD 550.3 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 240 |
| Tables, Charts & Figures: | 105 |
| Segments covered: | By Application, By Source |
| Companies covered:: | Grifols Revo Biologics Takeda Pharmaceutical Company Octapharma CSL Behring Kedrion biopharma LFB Lee BioSolutions Scripps Laboratories Endpoint Health Sanofi Sysmex Thermo Fisher Scientific Others Key Players |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
The North America Antithrombin Market Size is driven by rise in number of complex surgical procedures, particularly cardiovascular surgeries and organ transplants, where maintaining precise anticoagulation is critical. In these environments, antithrombin III acts as a necessary adjunct to heparin, especially in patients exhibiting heparin resistance. the region is experiencing a demographic shift toward an ageing population, which naturally correlates with a higher incidence of venous thromboembolism (VTE) and chronic kidney diseases that deplete natural anticoagulant levels.
Furthermore, the market is driven by enhanced diagnostic capabilities. The widespread adoption of advanced genetic screening for the SERPINC1 gene has led to a higher identification rate of hereditary antithrombin deficiencies, creating a consistent, long-term patient base for replacement therapies. From a technological standpoint, the transition from plasma-derived products to recombinant platforms, such as those utilizing transgenic animal milk, has bolstered supply chain stability and addressed safety concerns regarding blood-borne pathogens.
Restraining Factors
The antithrombin market in North America is constrained by the high cost of therapy, driven by the intricate biotechnological processes required for both plasma-derived and recombinant production. Furthermore, the stringent regulatory landscape enforced by the FDA necessitates prolonged clinical trials and high compliance expenditures, delaying the market entry of new biologics. Intense competition from alternative anticoagulants also hampers growth
Market Segmentation
The North America antithrombin market share is classified into application, source and thrombin type.
- The therapeutics segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
The North America Antithrombin Market Size is segmented by application into therapeutics, diagnostics, research, and others. Among these, the base metal smelters segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is due to the clinical necessity of antithrombin III in managing hereditary deficiencies and its critical role as an adjunct therapy for patients exhibiting heparin resistance during major cardiac surgeries, the high volume of inpatient surgical procedures in the U.S. ensures that therapeutic usage remains the primary revenue generator.
- The human derived segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period.
Based on source The North America Antithrombin Market Size is segmented into human derived, recombinant goat milk, and others. Among these, the human derived plasma segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. due to its long-standing history of clinical validation and established plasma fractionation infrastructure.
- The anticoagulant segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
The North America antithrombin market is segmented by thrombin type into anticoagulant, antiplatelets, thrombolytic drugs and others. Among these, the base metal smelters segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is due to the clinical necessity of antithrombin III in managing hereditary deficiencies and its critical role as an adjunct therapy for patients exhibiting heparin resistance during major cardiac surgeries, the high volume of inpatient surgical procedures in the U.S. ensures that therapeutic usage remains the primary revenue generator.
Competitive Analysis:
The report offers the appropriate analysis of the key organisations/companies involved within the North America antithrombin market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborate analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Grifols
- Revo Biologics
- Takeda Pharmaceutical Company
- Octapharma
- CSL Behring
- Kedrion biopharma
- LFB
- Lee BioSolutions
- Scripps Laboratories
- Endpoint Health
- Sanofi
- Sysmex
- Thermo Fisher Scientific
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Developments:
- In November 2025, Grifols launched THROMBATE III® (antithrombin III [human]) to include paediatric patients with hereditary antithrombin deficiency (hATd) after receiving expanded indication approval from United States Food and Drug Administration. It is the first and only antithrombin concentrate approved for both adults and children with this rare, high-risk blood clotting disorder
- In June 2025, Sysmex launched the CN-6000 automated coagulation analyser. This platform integrates the Innovance Antithrombin assay, streamlining the rapid detection of deficiency levels in clinical settings across the U.S. and Canada.
- In March 2025, Sanofi launched Qfitlia (fitusiran), a first-in-class siRNA therapy that works by lowering antithrombin levels to rebalance coagulation in patients with haemophilia A or B. While it functions by reducing antithrombin rather than replacing it, this is a major shift in how the antithrombin pathway is therapeutically manipulated in North America.
Market Segment
This study forecasts revenue at the North America, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the North America antithrombin market based on the below-mentioned segments:
North America Antithrombin Market, By Application
- Therapeutics
- Diagnostics
- Research
- Others
North America Antithrombin Market, By Source
- Human Derived
- Recombinant Goat Milk
- Others
North America Antithrombin Market, By Thrombin Type
- Anticoagulant
- Antiplatelets
- Thrombolytic Drugs
- Others
Frequently Asked Questions (FAQ)
-
What is the North America antithrombin market size?North America antithrombin market size is expected to grow from USD 310.3 million in 2024 to USD 550.3 million by 2035, growing at a CAGR of 5.35% during the forecast period 2025-2035.
-
What is the projected growth for the North America antithrombin market through 2035?The market is expected to grow from an estimated USD 310.3 million in 2024 to approximately USD 550.3 million by 2035. This represents a steady Compound Annual Growth Rate (CAGR) of 5.35% during the forecast period of 2025–2035.
-
What exactly is antithrombin, and why is it medically significant?Antithrombin III (ATIII) is a vital plasma protein that acts as a natural anticoagulant. It works by neutralizing thrombin and other clotting factors in the blood. It is essential for managing hereditary deficiencies and is often used during complex surgeries, such as cardiopulmonary bypasses, to prevent dangerous blood clots.
-
What are the main challenges or restraints facing the market?Growth is primarily hampered by the high cost of therapy resulting from complex biotechnological production. Additionally, the stringent FDA regulatory landscape requires long clinical trials, and there is intense competition from alternative, lower-cost anticoagulants
-
Is there any government support or funding available for this market?Yes. The market benefits from FDA Orphan Drug Designations and R&D tax credits, which help offset the costs of developing treatments for rare conditions. Furthermore, the National Institutes of Health (NIH) provides federal funding and clinical grants that support biotech firms in exploring new therapeutic indications.
Need help to buy this report?