North America Antiepileptic Drugs Market Size, Share, and COVID-19 Impact Analysis, By Product (First Generation, Second Generation, Third Generation), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, and Others), and North America Antiepileptic Drugs Market Insights, Industry Trend, Forecasts to 2035
Industry: HealthcareNorth America Antiepileptic Drugs Market Insights Forecasts to 2035
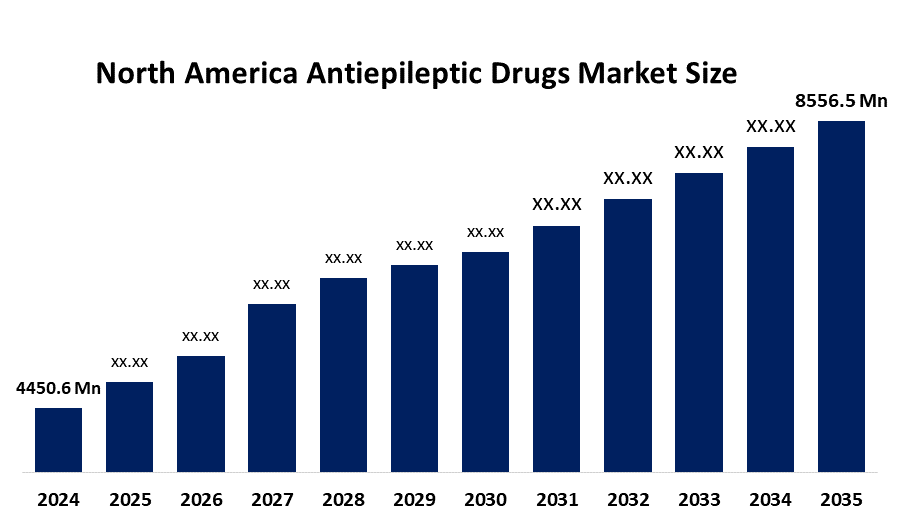
- The North America Antiepileptic Drugs Market Size Was Estimated at USD 4450.6 Million in 2024
- The Market Size is Expected to Grow at a CAGR of Around 6.12% from 2025 to 2035
- The North America Antiepileptic Drugs Market Size is Expected to Reach USD 8556.5 Million by 2035
Get more details on this report -
According to a research report published by Spherical Insights & Consulting, The North America Antiepileptic Drugs Market Size is anticipated to reach USD 8556.5 Million by 2035, Growing at a CAGR of 6.12% from 2025 to 2035. The antiepileptic drug market in North America is driven by rising prevalence of neurological disorders, massive investments in neurology, sustained funding from North American region governments to promote the research and development of novel drug delivery systems for neurological disorders and innovation in advanced diagnostics.
Market Overview
The North American antiepileptic drugs market represents the landscape for seizure management. This market involves the research and supply of diverse pharmacological treatments spanning traditional sodium channel blockers to modern synaptic vesicle modulators specifically designed to inhibit the abnormal neuronal firing associated with epilepsy and related neurological disorders.
The region’s market dominance is fuelled by a growing patient demographic, as nearly 3.4 million individuals in the U.S. alone require ongoing seizure therapy. This demand is further intensified by an ageing population more susceptible to strokes and traumatic brain injuries, both of which are common precursors to epilepsy. On the innovation front, the industry is moving toward precision neurology, focusing on third-generation medications that minimize cognitive side effects while maximizing seizure control. Recent breakthroughs in delivery methods, such as intranasal rescue therapies and extended-release capsules, have also revolutionized patient compliance and emergency intervention.
Initiatives like the National Plan for Epilepsy and sustained funding from the NIH and CDC facilitate the transition of laboratory research into clinical practice. Furthermore, FDA incentives for orphan drug development have accelerated the arrival of treatments for rare, treatment-resistant paediatric syndromes. These policy-driven advancements, paired with comprehensive insurance coverage across the U.S. and Canada, ensure North America remains the global hub for epilepsy care innovation.
Report Coverage
This research report categorizes the market for the North America antiepileptic drugs based on various segments and regions, and forecasts revenue growth and analyses trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the North America antiepileptic drugs market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the North America antiepileptic drugs market.
North America Antiepileptic Drugs Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 4450.6 Million |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | CAGR of 6.12% |
| 2035 Value Projection: | USD 8556.5 |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 210 |
| Tables, Charts & Figures: | 95 |
| Segments covered: | By Product, By Distribution Channel |
| Companies covered:: | UCB, Pfizer, GlaxoSmithKline, Novartis, Immedica, Lupin Ltd, Jazz Pharmaceuticals, Supernus Pharmaceuticals, Xenon Pharmaceuticals, Teva Pharmaceutical, Otsuka Pharmaceuticals, Elsai Ltd, Others, and Key Players |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
The North America antiepileptic drugs market is driven by the increasing prevalence of epilepsy, which currently impacts approximately 3.4 million people in the U.S. alone. This burden is intensified by a rapidly ageing population; as individuals over 65 become a larger demographic, the incidence of seizures related to strokes and neurodegenerative conditions has risen sharply. Increased public awareness and the integration of AI-driven diagnostic tools have led to earlier and more accurate diagnoses, ensuring that a larger patient pool enters the long-term medication cycle. Technological innovation also plays a critical role in the growth of this market, with the discovery and development of novel delivery methods, such as intranasal rescue therapies and extended-release formulations has significantly improved patient compliance.
Restraining Factors
The North America antiepileptic drugs are constrained by factors such as high costs of novel drugs, significant side effects, including cognitive impairment, dizziness, and mood disturbances. Stringent FDA safety warnings, and the high rate of drug-resistant (refractory) epilepsy limit the long-term usage and market penetration of existing therapies.
Market Segmentation
The North America anti-epileptic drugs market share is classified into product and distribution channel.
- The second-generation segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
The North America antiepileptic drugs market is segmented by product into first generation, second generation, and third generation. Among these, the second-generation segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. This is due to fewer drug-drug interactions and better safety profiles (less toxicity to the liver). They are effective against multiple seizure types, making them the primary choice for initial monotherapy in North American hospitals and clinics.
- The hospital pharmacies segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period.
The North America antiepileptic drugs market is segmented by distribution channel into hospital pharmacies, retail pharmacies, and others. Among these, the hospital pharmacies segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. In North America, the majority of epilepsy diagnoses and acute seizure events are managed in hospital settings. Hospitals are the primary sites for administering intravenous (IV) formulations of AEDs, which are significantly more expensive than oral versions. Specialised epilepsy centres in the U.S. and Canada are often housed within hospitals, meaning high-cost third-generation drugs are initially prescribed and dispensed here.
Competitive Analysis:
The report offers the appropriate analysis of the key organisations/companies involved within the North America antiepileptic drugs market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborate analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- UCB
- Pfizer
- GlaxoSmithKline
- Novartis
- Immedica
- Lupin Ltd
- Jazz Pharmaceuticals
- Supernus Pharmaceuticals
- Xenon Pharmaceuticals
- Teva Pharmaceutical
- Otsuka Pharmaceuticals
- Elsai Ltd
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Developments:
- In February 2026, Lupin Limited launched the first generic version of Brivaracetam Oral Solution in the U.S., following FDA approval, a third-generation antiepileptic drug.
- In February 2025, Immedica Pharma completed the acquisition of Marinus Pharmaceuticals for USD 151 million, consolidating the rare-epilepsy market in North America, bringing the commercialisation of ganaxolone under Immedica's specialized neurology umbrella.
- In October 2024, Alkem Laboratories received final FDA approval and launched Topiramate Oral Solution (generic Eprontia), providing a new liquid formulation for paediatric and geriatric populations with swallowing difficulties.
Market Segment
This study forecasts revenue at the Indonesia, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the North America antiepileptic drug market based on the below-mentioned segments:
North America Antiepileptic Drugs Market, By Product
- First Generation,
- Second Generation
- Third Generation
North America Antiepileptic Drugs Market, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Others
Frequently Asked Questions (FAQ)
-
Q: What is the North America anti-epileptic drugs market size?A: The North America antiepileptic drugs market size is expected to grow from USD 4450.6 million in 2024 to USD 8556.5 million by 2035, growing at a CAGR of 6.12% during the forecast period 2025-2035.
-
Q: Who are the target audiences for this market report?A: The report targets market players, investors, end-users, government authorities, consulting and research firms, venture capitalists, and value-added resellers (VARs).
Need help to buy this report?