Global Lipid Nanoparticle CDMO Market Size, Share, and COVID-19 Impact Analysis, By Service Type (Development & Process Development, LNP Formulation & Encapsulation, Analytical & Quality Control, cGMP Drug Product Manufacturing, and Fill-Finish Lyophilization), By Application (Infectious Disease Vaccines, Oncology Therapeutics, Rare/Genetic & Metabolic Disorders, and Other Therapeutics), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 - 2035
Industry: HealthcareGlobal Lipid Nanoparticle CDMO Market Size Insights Forecasts to 2035
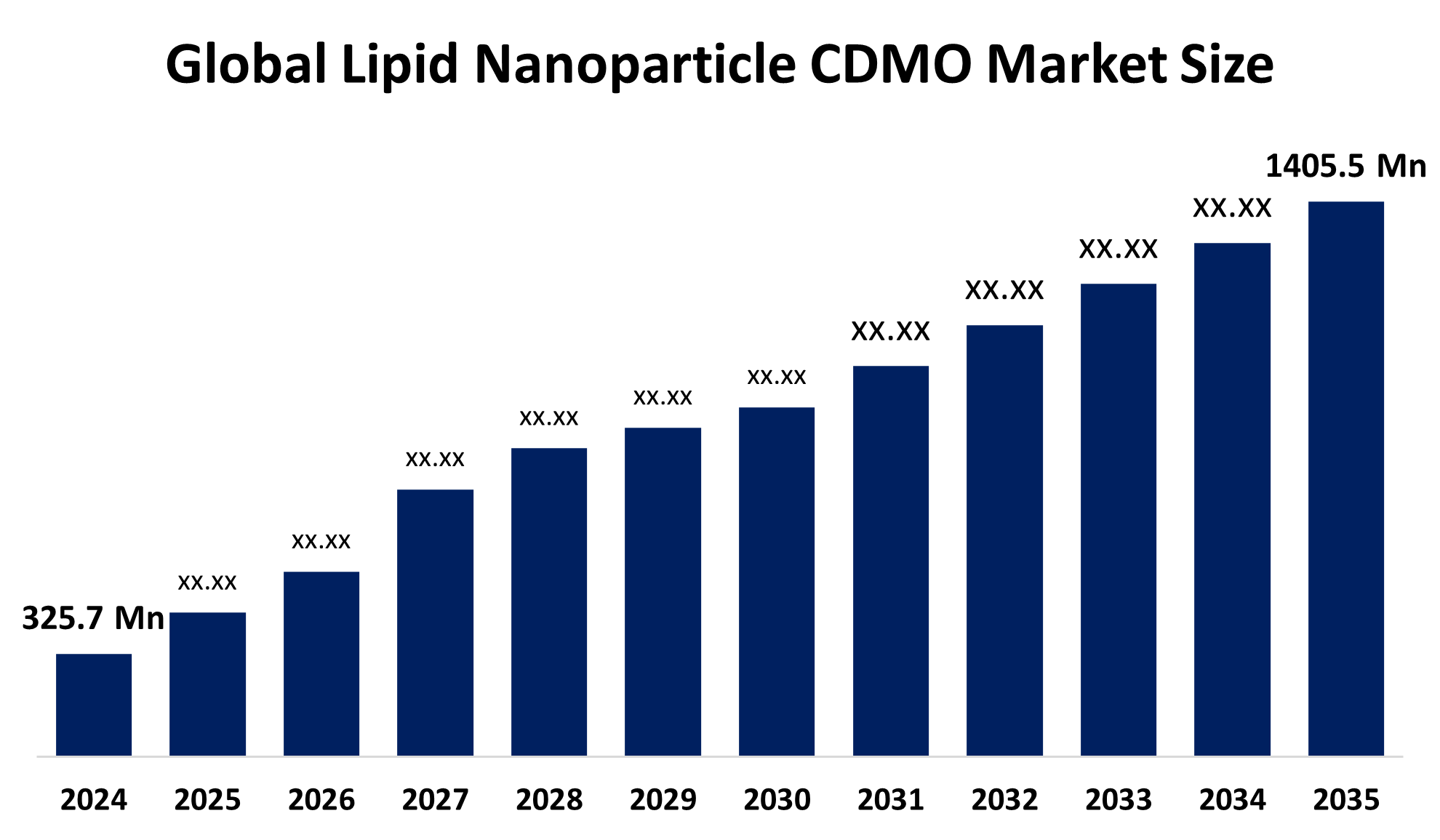
- The Global Lipid Nanoparticle CDMO Market Size Was Estimated at USD 325.7 Million in 2024
- The Market Size is Expected to Grow at a CAGR of around 14.22% from 2025 to 2035
- The Worldwide Lipid Nanoparticle CDMO Market Size is Expected to Reach USD 1405.5 Million by 2035
- Asia Pacific is expected to grow the fastest during the forecast period.
Get more details on this report -
According to a research report published by Spherical Insights and Consulting, The Global Lipid Nanoparticle CDMO Market Size was worth around USD 325.7 Million in 2024 and is predicted to grow to around USD 1405.5 Million by 2035 with a Compound Annual Growth Rate (CAGR) of 14.22% from 2025 and 2035. The market for lipid nanoparticle CDMO offers several opportunities to grow due to the development of ionizable lipids for LNP formulations. Furthermore, the expanding use of mRNA vaccines, not only for COVID-19 but also in oncology, rare diseases, and gene therapy, is promoting the industry for lipid nanoparticles.
Market Overview
The global industry of lipid nanoparticle CDMO encompasses specialized service providers developing and manufacturing LNPs for pharmaceutical companies, which is driven by the surging demand for advanced delivery systems in mRNA vaccines, oncology, and gene therapies. A lipid nanoparticle CDMO (contract development and manufacturing organization) provides specialized services for designing, formulating, analyzing, and manufacturing lipid-based delivery vehicles for mRNA, siRNA, and other nucleic acid therapeutics. The growing trend of biopharma businesses outsourcing formulation and manufacturing services contributes to this rise. Furthermore, it is anticipated that continued developments in nanotechnology and the growing uses of LNPs in cancer and gene therapy quicken market growth.
Innovation and market expansion are anticipated as a result of major players' growing R&D expenditures and expanding partnerships. For instance, in march 2026, BioMed Realty and CordenPharma Colorado teamed up for the expansion of the Peptide Development Lab in a new LEED Gold-certified facility at Flatiron Park in Boulder.
Report Coverage
This research report categorizes the lipid nanoparticle CDMO market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the lipid nanoparticle CDMO market. Recent market developments and competitive strategies such as expansion, type launch, development, partnership, merger, and acquisition have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the lipid nanoparticle CDMO market.
Global Lipid Nanoparticle CDMO Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 325.7 Million |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | 14.22% |
| 2035 Value Projection: | USD 1405.5 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 225 |
| Tables, Charts & Figures: | 102 |
| Segments covered: | By Service Type, By Application |
| Companies covered:: | AGC Biologics, CordenPharma, Curia, Eurogentec, Evonik Health Care, Fujifilm Toyama Chemical, Lonza, PCI Pharma Services, Polymun Scientific, Porton Advanced, Recipharm, Rentschler Biopharma, Samsung Biologics, Thermo Fisher Scientific and Other Key Players |
| Pitfalls & Challenges: | and COVID-19 Impact Analysis |
Get more details on this report -
Driving Factors
The lipid nanoparticle CDMO is driven by pipeline expansion in mRNA vaccines and therapeutics beyond COVID 19 to target a broad range of communicable and non-communicable diseases. Support for government-backed investments and strategic partnerships is surging for mRNA vaccines and therapeutic production, which contributes to propelling market growth. Outsourcing RNA-LNP production for streamlining manufacturing processes, reducing operational complexities, and enhancing cost efficiency are contributing to escalating market growth.
Restraining Factors
The Global Lipid Nanoparticle CDMO Market is restricted by factors like LNP IP litigation & licensing complexity, as well as GMP-grade lipid & critical component supply constraints. Further, the increased, complex manufacturing costs, and lack of specialized technical expertise are challenging the market.
Market Segmentation
The lipid nanoparticle CDMO market share is classified into service type and application.
- The development & process development segment dominated the market with the largest share of around 35.2% in 2024 and is projected to grow at a substantial CAGR during the forecast period.
Based on the service type, the Global Lipid Nanoparticle CDMO Market is divided into development & process development, LNP formulation & encapsulation, analytical & quality control, cGMP drug product manufacturing, and fill-finish lyophilization. Among these, the development & process development segment dominated the market with the largest share of around 35.2% in 2024 and is projected to grow at a substantial CAGR during the forecast period. Lipid nanoparticle CDMOs provide end-to-end development, process optimization, and cGMP manufacturing for advanced therapeutics (mRNA, siRNA). Formulation screening, microfluidic parameter tuning, scale-up, and analytical characterization. cGMP drug-product manufacturing remains the largest pool of approved vaccines and therapeutics.
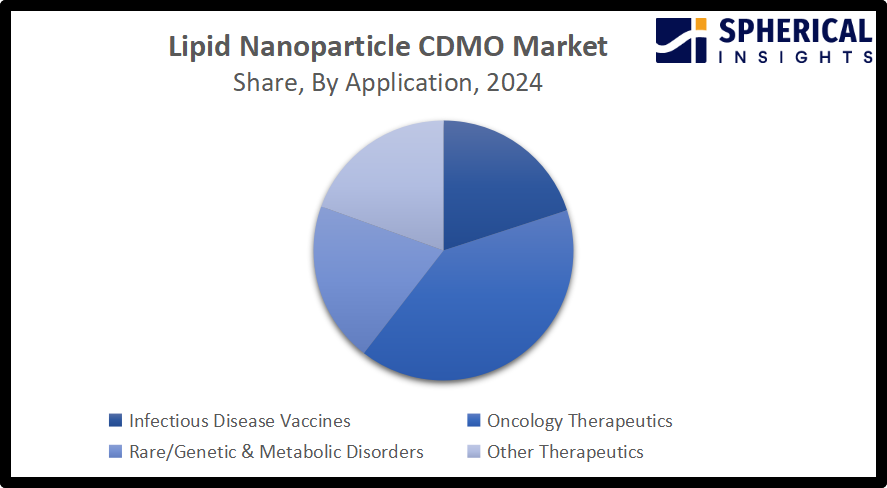
- The oncology therapeutics segment accounted for the largest share of around 40.5% in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the application, the Global Lipid Nanoparticle CDMO Market is divided into infectious disease vaccines, oncology therapeutics, rare/genetic & metabolic disorders, and other therapeutics. Among these, the oncology therapeutics segment accounted for the largest share of around 40.5% in 2024 and is anticipated to grow at a significant CAGR during the forecast period. An ongoing advancement in nanotechnology is contributing to driving the market in the oncology therapeutics segment. CDMOs provide important expertise in formulation development, process scaling, and GMP-compliant manufacturing for complex, 10-1000nm, lipid-based systems that include components like mRNA, siRNA, and CRISPR components to specific tumor sites.
Get more details on this report -
Regional Segment Analysis of the Lipid Nanoparticle CDMO Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
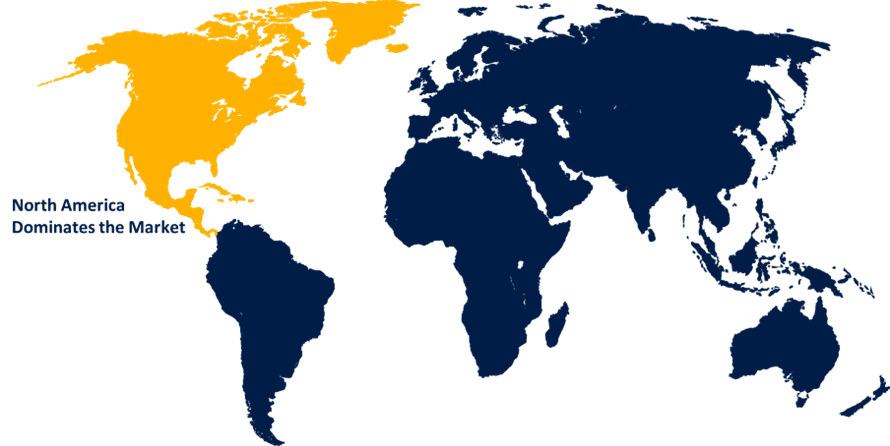
North America is anticipated to hold the largest share of the lipid nanoparticle CDMO market over the predicted timeframe.
Get more details on this report -
North America is anticipated to hold the largest share of about 45.5% in the lipid nanoparticle CDMO market over the predicted timeframe. The market ecosystem in North America is strong, with the surging innovation in research and formulation activities. The demand for lipid nanoparticle CDMO has been driven by the region's increasing demand for mRNA-based vaccines and therapeutics, along with an increased prevalence of chronic/infectious diseases requiring targeted delivery and R&D investment. The U.S. accounts for the largest portion of about 43.0% share within the North America region for the lipid nanoparticle CDMO market, driven by the presence of a strong biopharmaceutical ecosystem, investments in mRNA/LNP technologies, and a supportive regulatory environment and funding programs.
Asia Pacific is expected to grow at a rapid CAGR of around 15.3% in the lipid nanoparticle CDMO market during the forecast period. The Asia Pacific area has a thriving market for lipid nanoparticle CDMO due to its rising healthcare investments and expanding biopharmaceutical manufacturing capabilities, advanced research facilities in countries like Japan and South Korea. Due to their governments actively supporting the regulatory frameworks, and long-term funding is supporting the market for lipid nanoparticle CDMO. Further, the presence of specialized and large-scale contract manufacturers like Samsung Biologics, WuXi AppTec/WuXi Biologics, FUJIFILM Corporation, and NOF Corporation also plays a major role in the region's market. China is leading the Asia Pacific lipid nanoparticle CDMO market, with about 15.0% CAGR value, owing to the increasing investment in mRNA and nucleic acid therapeutics.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the Global Lipid Nanoparticle CDMO Market, along with a comparative evaluation primarily based on their type of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes type development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- AGC Biologics
- CordenPharma
- Curia
- Eurogentec
- Evonik Health Care
- Fujifilm Toyama Chemical
- Lonza
- PCI Pharma Services
- Polymun Scientific
- Porton Advanced
- Recipharm
- Rentschler Biopharma
- Samsung Biologics
- Thermo Fisher Scientific
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting And Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In February 2026, Contract development and manufacturing organization (CDMO) Cellipont Bioservices announced a partnership with Soter Bio, a CDMO specializing in the development of lipid nanoparticles for RNA therapies. The companies would collaborate on integrated manufacturing techniques to produce complex cell therapies.
- In September 2025, Evonik, a global specialty chemicals company, and Ethris, a clinical-stage biotechnology company pioneering next-generation RNA therapeutics and vaccines, entered a strategic collaboration to develop and market a novel lipid nanoparticle (LNP) platform for nucleic acid delivery.
- In March 2025, Amaran Biotechnology Inc., a subsidiary of OBI Pharma specialising in Contract Development and Manufacturing Organization (CDMO) services for next-generation pharmaceuticals, signed a Memorandum of Understanding (MoU) with Nippon Fine Chemical Co., Ltd., and its Taiwan subsidiary, Zillion Fine Chemicals International Co., Ltd.
- In July 2024, Agilent is upping CDMO presence through $925m Biovectra buy. The deal announced today sees Agilent Technologies bolster its current oligonucleotide and CRISPR-based therapeutics manufacturing services with the addition of contract development and manufacturing organization (CDMO) Biovectra.
- In June 2024, CordenPharma partnered with Certest to develop Ionizable Lipids for LNP formulations. Switzerland-based CordenPharma, a contract development and manufacturing organization, announced that it has partnered with Certest, a Spain-based company focused on lipid nanoparticle (LNP)-based drug delivery and API synthesis, to develop a portfolio of ionizable lipids for LNP formulations.
- In May 2023, Aldevron, a subsidiary of Danaher, unveiled plans to expand its mRNA manufacturing operations to include lipid nanoparticle (LNP) encapsulation and aseptic fill-finish capabilities.
- In January 2022, Merck KGaA, Darmstadt, Germany, a leading science and technology company, announced the signing of a definitive agreement to acquire Exelead, a biopharmaceutical contract development and manufacturing organization (CDMO), for approximately USD 780 million in cash.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the Global Lipid Nanoparticle CDMO Market based on the below-mentioned segments:
Global Lipid Nanoparticle CDMO Market, By Service Type
- Development & Process Development
- LNP Formulation & Encapsulation
- Analytical & Quality Control
- cGMP Drug Product Manufacturing
- Fill-Finish Lyophilization
Global Lipid Nanoparticle CDMO Market, By Application
- Infectious Disease Vaccines
- Oncology Therapeutics
- Rare/Genetic & Metabolic Disorders
- Other Therapeutics
Global Lipid Nanoparticle CDMO Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
-
1.What is the market size of the lipid nanoparticle CDMO market?The global lipid nanoparticle CDMO market size is expected to grow from USD 325.7 Million in 2024 to USD 1405.5 Million by 2035, at a CAGR of 14.22% during the forecast period 2025-2035.
-
2.Which region holds the largest share of the lipid nanoparticle CDMO market?North America is anticipated to hold the largest share of the lipid nanoparticle CDMO market over the predicted timeframe.
-
3.What is the forecasted CAGR of the Global Lipid nanoparticle CDMO Market from 2024 to 2035?The market is expected to grow at a CAGR of around 14.22% during the period 2024–2035.
-
4.Who are the top companies that are involved in the Global Lipid Nanoparticle CDMO Market?Key players include AGC Biologics, CordenPharma, Curia, Eurogentec, Evonik Health Care, Fujifilm Toyama Chemical, Lonza, PCI Pharma Services, Polymun Scientific, Porton Advanced, Recipharm, Rentschler Biopharma, Samsung Biologics, and Thermo Fisher Scientific.
-
5.What are the main drivers in the lipid nanoparticle CDMO market?A pipeline expansion in mRNA vaccines and therapeutics, government-backed investments and strategic partnerships, and the need to streamline manufacturing processes, reducing operational complexities, and enhancing cost efficiency of production are major market growth drivers of the lipid nanoparticle CDMO market.
-
6.What challenges are limiting the adoption of lipid nanoparticle CDMO treatment?Factors like increased, complex manufacturing costs, lack of specialized technical expertise, and regulatory complexities remain key restraints in the lipid nanoparticle CDMO market.
-
7.What are the key trends in the lipid nanoparticle CDMO market?The development of ionizable lipids for LNP formulations and expanding mRNA therapeutics for oncology & gene therapy are key trends in the lipid nanoparticle CDMO market.
Need help to buy this report?