Japan Transcatheter Heart Valve Market Size, Share, By Technology (Balloon Expanded Transcatheter Valve and Self-Expanded Transcatheter Valve), By Application (Transcatheter Aortic Valve, Transcatheter Pulmonary Valve, and Transcatheter Mitral Valve), Japan Transcatheter Heart Valve Market Industry Trend, Forecasts to 2035.
Industry: HealthcareJapan Transcatheter Heart Valve Market Insights Forecasts to 2035
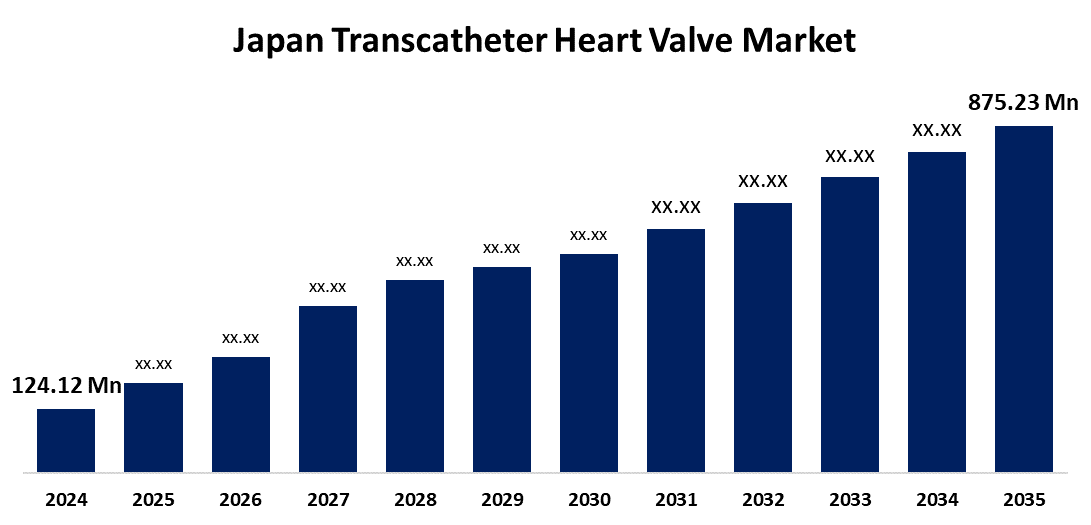
- Japan Transcatheter Heart Valve Market Size 2024: USD 124.12 million
- Japan Transcatheter Heart Valve Market Size 2035: USD 875.23 million
- Japan Transcatheter Heart Valve Market CAGR 2024: 19.43%
- Japan Transcatheter Heart Valve Market Segments: Technology and Applications.
Get more details on this report -
The Japan transcatheter heart valve market includes minimally invasive heart valve replacement and repair devices, such as transcatheter aortic, mitral, and pulmonary valves. These devices are delivered via catheter (without open-chest surgery) to treat valvular heart diseases, especially in elderly and high-risk patients. They improve cardiac function and reduce surgical trauma compared to traditional open procedures. Furthermore, the Japan transcatheter heart valve market growth is fueled by Japan’s ageing population, increasing prevalence of aortic stenosis and other valvular heart diseases, and rising preference for minimally invasive procedures. Advancements in valve technology, improved clinical outcomes, shorter hospital stays, and growing physician expertise accelerate adoption. Government support and reimbursement policies for innovative cardiac therapies also contribute to strong market expansion.
The Japan government, through the Pharmaceuticals and Medical Devices Agency (PMDA) and the Ministry of Health, Labour and Welfare (MHLW), facilitates the approval and reimbursement of transcatheter heart valves. Policies emphasise patient safety, clinical efficacy, and cost-effectiveness. Conditional approvals, universal healthcare coverage, and support for innovative cardiovascular technologies encourage adoption, especially for elderly or high-risk patients needing minimally invasive interventions.
Japan transcatheter heart valve market trends include growing adoption of TAVR procedures, development of next-generation transcatheter valves with improved durability, and hybrid operating rooms. Use of advanced imaging for precise implantation is rising. Mitral and pulmonary valve replacements are emerging alongside aortic valve procedures. Telehealth integration and early detection of valvular heart diseases are boosting patient referrals and procedural volume.
Market Dynamics of the Japan Transcatheter Heart Valve Market:
The Japan transcatheter heart valve market is driven by increasing cardiovascular disease prevalence, the ageing population, and patient preference for minimally invasive procedures. Technological innovations in valve design and delivery systems, reduced complications, shorter recovery, and improved long-term outcomes strengthen adoption. Physician training programs and hospital infrastructure upgrades also support market growth, enabling wider availability of transcatheter heart valve procedures across Japan.
The Japan transcatheter heart valve market restraints include high device costs, the need for specialised surgical expertise, and complex procedural requirements. Regulatory approval processes, reimbursement limitations, and hospital infrastructure constraints can delay adoption. Competition from traditional surgical valves, lack of awareness among patients and some clinicians, and stringent safety requirements also limit market penetration. Smaller hospitals may struggle to implement advanced transcatheter procedures.
The opportunities exist in expanding indications to mitral and pulmonary valves, next-generation valve designs, and hybrid surgical suite adoption. Early diagnosis programs, outpatient transcatheter procedures, and partnerships between global and domestic manufacturers offer growth potential. Rising patient awareness, increased funding for minimally invasive cardiac therapies, and advanced imaging technologies further enhance the market for innovative transcatheter heart valve solutions.
Japan Transcatheter Heart Valve Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 124.12 Million |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | 19.43 % |
| 2035 Value Projection: | USD 875.23 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 220 |
| Tables, Charts & Figures: | 88 |
| Segments covered: | By Technology, By Application |
| Companies covered:: | Edwards Lifesciences, Medtronic Plc, Abbott Laboratories, Boston Scientific Corporation, LivaNova PLC, MicroPort Scientific Corporation, Meril Life Sciences, Venus Medtech (Hangzhou) Inc., Artivion, Inc., and other |
| Pitfalls & Challenges: | COVID-19 Impact, Challenges, Future, Growth, & Analysis |
Get more details on this report -
Market Segmentation
The Japan transcatheter heart valve market share is classified into technology and applications.
By Technology:
The Japan transcatheter heart valve market is divided by technology into balloon-expanded transcatheter valve and self-expanded transcatheter valve. Among these, the balloon-expanded transcatheter valve segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. The balloon-expanded transcatheter valve segment dominates due to its offer of precise placement, strong clinical evidence, and reliable outcomes in aortic valve replacement procedures. Japanese hospitals and cardiologists are highly experienced with this technology, increasing confidence and adoption. Their proven safety profile and consistent performance make them the preferred choice over self-expanding valves in most cases.
By Application:
The Japan transcatheter heart valve market is divided by application into transcatheter aortic valve, transcatheter pulmonary valve, and transcatheter mitral valve. Among these, the transcatheter aortic valve segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. The transcatheter aortic valve industry dominates because aortic stenosis is highly prevalent among Japan’s ageing population. TAVR procedures are widely accepted due to strong clinical evidence, high success rates, and reduced recovery time compared to open surgery. Established reimbursement policies and extensive physician experience further support higher procedural volumes than mitral or pulmonary valve interventions.
Competitive Analysis:
The report offers the appropriate analysis of the key organisations/companies involved within the Japan transcatheter heart valve market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborate analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in Japan Transcatheter Heart Valve Market:
- Edwards Lifesciences
- Medtronic Plc
- Abbott Laboratories
- Boston Scientific Corporation
- LivaNova PLC
- MicroPort Scientific Corporation
- Meril Life Sciences
- Venus Medtech (Hangzhou) Inc.
- Artivion, Inc.
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the Japan, regional, and country levels from 2020 to 2035. Decisions advisors has segmented the Japan transcatheter heart valve market based on the below-mentioned segments:
Japan Transcatheter Heart Valve Market, By Technology
- Balloon Expanded Transcatheter Valve
- Self-Expanded Transcatheter Valve
Japan Transcatheter Heart Valve Market, By Application
- Transcatheter Aortic Valve
- Transcatheter Pulmonary Valve
- Transcatheter Mitral Valve
Frequently Asked Questions (FAQ)
-
What is the Japan transcatheter heart valve market size?Japan transcatheter heart valve market is expected to grow from USD 124.12 million in 2024 to USD 875.23 million by 2035, growing at a CAGR of 19.43 % during the forecast period 2025-2035.
-
What are the key growth drivers of the Japan transcatheter heart valve market?The Japan Transcatheter Heart Valve Market includes a growing Aging population, rising aortic stenosis cases, preference for minimally invasive procedures, technological advancements, improved outcomes, and supportive reimbursement policies.
-
What factors restrain the Japan Transcatheter Heart Valve Market?The Japan Transcatheter Heart Valve Market is restrained by High device costs, complex procedures, the need for specialised expertise, strict regulatory approvals, reimbursement limitations, and infrastructure gaps in smaller hospitals.
-
How is the Japan transcatheter heart valve market segmented by application?The Japan transcatheter heart valve market is segmented into transcatheter aortic valve, transcatheter pulmonary valve, and transcatheter mitral valve
-
Who are the key players in the Japan Transcatheter Heart Valve Market?Key companies in the Japan Transcatheter Heart Valve Market include Edwards Lifesciences, Medtronic Plc, Abbott Laboratories, Boston Scientific Corporation, LivaNova PLC, MicroPort Scientific Corporation, Meril Life Sciences, Venus Medtech (Hangzhou) Inc., Artivion, Inc., and other
Need help to buy this report?