Japan Pharmaceutical Microbiology QC Testing Market Size, Share, By Product (Instruments, Reagents & Kits, Accessories, and Software), By Test Type (Traditional/Conventional Testing and Rapid Testing), By Technique (Growth-based Testing, Nucleic Acid-based Testing, Cellular Component-based Testing, Viability-based Testing, and Others), By Application (Bioburden Testing, Sterility Testing, Environmental Monitoring, Endotoxin Testing, Pyrogen Testing, Mycoplasma Testing, Microbial Identification, and Others), Japan Pharmaceutical Microbiology QC Testing Market Insights, Industry Trend, Forecasts to 2035.
Industry: HealthcareJapan Pharmaceutical Microbiology QC Testing Market Insights Forecasts to 2035
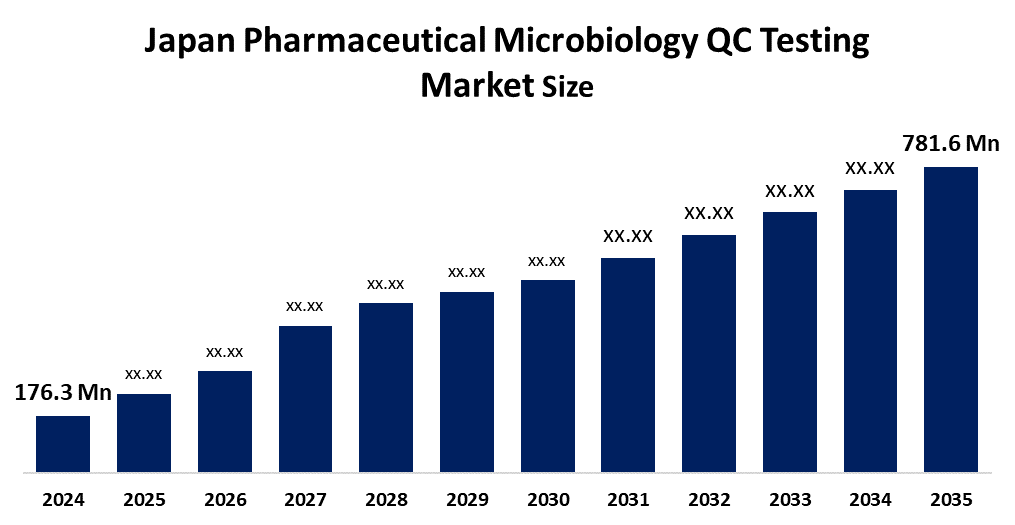
- Japan Pharmaceutical Microbiology QC Testing Market Size 2024: USD 176.3 Mn
- Japan Pharmaceutical Microbiology QC Testing Market Size 2035: USD 781.6 Mn
- Japan Pharmaceutical Microbiology QC Testing Market CAGR 2024: 14.5%
- Japan Pharmaceutical Microbiology QC Testing Market Segments: Product, Test Type, Technique, and Application
Get more details on this report -
The Japan pharmaceutical microbiology QC testing market comprises instruments, reagents, and analytical systems used to detect and control microbial contamination in pharmaceutical manufacturing environments. The solutions provide support for sterility assurance bioburden testing endotoxin detection and microbial identification combined with environmental monitoring. The production process depends on these materials for creating injectables biologics vaccines ophthalmic drugs and advanced therapeutic products. The market expansion occurs because Japanese pharmaceutical companies maintain strict GMP compliance standards and increase their biologics manufacturing facilities.
Japan depends on imported rapid microbial detection platforms, PCR-based systems, and specialized endotoxin testing reagents to meet advanced quality standards. The introduction of automatic culture systems, continuous microbial observation tools, ATP-based quick testing systems, and digital laboratory data integration techniques is driving technological progress because these systems enable faster product release. The Pharmaceuticals and Medical Devices Act (PMD Act) and strict GMP enforcement by the PMDA mandate validated sterility and contamination control protocols across manufacturing facilities. The development of cell and gene therapy production facilities and the rise of aseptic manufacturing investment and the modernization of existing QC laboratories and the implementation of quick microbiological testing methods to enhance batch release processes provide companies with new business opportunities.
Market Dynamics of the Japan Pharmaceutical Microbiology QC Testing Market:
The Japan pharmaceutical microbiology QC testing market is driven by stringent GMP compliance requirements, increasing biologics and injectable drug production, and strict sterility assurance mandates across manufacturing facilities. Rising investments in aseptic processing lines, cleanroom expansion, and contamination control systems are strengthening demand for validated microbiological testing solutions. The growing adoption of rapid microbiological methods to shorten batch-release timelines further supports market expansion.
The Japan pharmaceutical microbiology QC testing market faces restraints due to high capital costs associated with advanced rapid testing platforms and automated systems. Lengthy validation procedures, regulatory approval timelines for new testing technologies, and the need for skilled microbiologists also limit faster adoption. Smaller manufacturers often rely on conventional culture-based methods due to budget and compliance complexities.
The Japan pharmaceutical microbiology QC testing market presents significant future opportunities through expansion of cell and gene therapy manufacturing, continuous production models, and high-value biologics pipelines. Increasing digitalization of quality control laboratories and integration of AI-based data monitoring systems are expected to enhance efficiency and traceability. Modernization of legacy testing infrastructure across domestic pharmaceutical plants will further accelerate demand for innovative microbiology QC solutions.
Japan Pharmaceutical Microbiology QC Testing Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 176.3 Million |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | 14.5% |
| 2035 Value Projection: | USD 781.6 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 210 |
| Tables, Charts & Figures: | 90 |
| Segments covered: | By Product, By Test Type, By Technique, By Application |
| Companies covered:: | Thermo Fisher Scientific Inc., bioMérieux SA, Charles River Laboratories International, Inc., Merck KGaA, Becton, Dickinson and Company (BD), Danaher Corporation, Sartorius AG, Rapid Micro Biosystems, Inc., Shimadzu Corporation, FUJIFILM Wako Pure Chemical Corporation, and Sysmex Corporation. |
| Pitfalls & Challenges: | COVID-19 Impact, Challenges, Future, Growth, & Analysis |
Get more details on this report -
Market Segmentation
The Japan pharmaceutical microbiology QC testing market share is classified into product, test type, technique, and application.
By Product:
The Japan pharmaceutical microbiology QC testing market is divided by product into instruments, reagents & kits, accessories, and software. Among these, the reagents & kits segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. The reagents & kits segment dominates due to recurring consumption in sterility, bioburden, and endotoxin testing, strict batch-release requirements, high replacement frequency, validation-specific formulations, and continuous demand across every production cycle within pharmaceutical quality control laboratories.
By Test Type:
The Japan pharmaceutical microbiology QC testing market is divided by test type into traditional/conventional testing and rapid testing. Among these, the rapid testing segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. The rapid testing segment dominates due to shorter turnaround times, faster batch-release decisions, reduced quarantine costs, integration with automated systems, compliance with evolving pharmacopeial standards, and increasing adoption of PCR- and ATP-based microbial detection methods in pharmaceutical manufacturing facilities.
By Technique:
The Japan pharmaceutical microbiology QC testing market is divided by technique into growth-based testing, nucleic acid-based testing, cellular component-based testing, viability-based testing, and others. Among these, the growth-based testing segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. The growth-based testing segment dominates because pharmacopeial sterility and microbial limit tests mandate culture-based confirmation, established validation protocols, regulatory familiarity, broad microorganism detection capability, and standardized incubation procedures widely embedded within Japanese pharmaceutical quality control frameworks.
By Application:
The Japan pharmaceutical microbiology QC testing market is divided by application into bioburden testing, sterility testing, environmental monitoring, endotoxin testing, pyrogen testing, mycoplasma testing, microbial identification, and others. Among these, the sterility testing segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. The sterility testing segment dominates due to mandatory release testing for injectable drugs, biologics, and ophthalmic formulations, zero-tolerance contamination thresholds, stringent cleanroom validation protocols, high testing frequency per production batch, and regulatory enforcement under Japanese pharmaceutical GMP standards.
Competitive Analysis:
The report offers the appropriate analysis of the key organisations/companies involved within the Japan pharmaceutical microbiology QC testing market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in Japan Pharmaceutical Microbiology QC Testing Market:
- Thermo Fisher Scientific Inc.
- bioMerieux SA
- Charles River Laboratories International, Inc.
- Merck KGaA
- Becton, Dickinson and Company (BD)
- Danaher Corporation
- Sartorius AG
- Rapid Micro Biosystems, Inc.
- Shimadzu Corporation
- FUJIFILM Wako Pure Chemical Corporation
- Sysmex Corporation
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the Japan, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the Japan pharmaceutical microbiology QC testing market based on the below-mentioned segments:
Japan Pharmaceutical Microbiology QC Testing Market, By Product
- Instruments
- Reagents & Kits
- Accessories
- Software
Japan Pharmaceutical Microbiology QC Testing Market, By Test Type
- Traditional/Conventional Testing
- Rapid Testing
Japan Pharmaceutical Microbiology QC Testing Market, By Technique
- Growth-based Testing
- Nucleic Acid-based Testing
- Cellular Component-based Testing
- Viability-based Testing
- Others
Japan Pharmaceutical Microbiology QC Testing Market, By Application
- Bioburden Testing
- Sterility Testing
- Environmental Monitoring
- Endotoxin Testing
- Pyrogen Testing
- Mycoplasma Testing
- Microbial Identification
- Others
Frequently Asked Questions (FAQ)
-
What is the Japan pharmaceutical microbiology QC testing market size?Japan pharmaceutical microbiology QC testing market is expected to grow from USD 176.3 million in 2024 to USD 781.6 million by 2035, growing at a CAGR of 14.5% during the forecast period 2025-2035.
-
What are the key growth drivers of the market?Market growth is driven by stringent GMP compliance requirements, increasing biologics and injectable drug production, rising investments in aseptic manufacturing, and growing adoption of rapid microbiological testing methods to shorten batch-release timelines.
-
What factors restrain the Japan pharmaceutical microbiology QC testing market?Key restraints include high capital investment required for advanced rapid testing systems, lengthy validation and regulatory approval procedures, and limited availability of skilled microbiology professionals in pharmaceutical quality control laboratories.
-
Who are the key players in the Japan pharmaceutical microbiology QC testing market?Key companies include Thermo Fisher Scientific Inc., bioMérieux SA, Charles River Laboratories International, Inc., Merck KGaA, Becton, Dickinson and Company (BD), Danaher Corporation, Sartorius AG, Rapid Micro Biosystems, Inc., Shimadzu Corporation, FUJIFILM Wako Pure Chemical Corporation, and Sysmex Corporation.
-
Who are the target audiences for this market report?The report targets market players, investors, end-users, government authorities, consulting and research firms, venture capitalists, and value-added resellers (VARs).
Need help to buy this report?