Global Ebola Vaccine Market Size, Share, and COVID-19 Impact Analysis, By Vaccine Type (Recombinant Vector Vaccine, Inactivated Virus Vaccine, DNA Vaccine, Subunit Vaccine, and Others), By End User (Hospitals, Government Health Departments, Research Institutes, NGOs, and Others), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 - 2035
Industry: HealthcareGlobal Ebola Vaccine Market Insights Forecasts to 2035
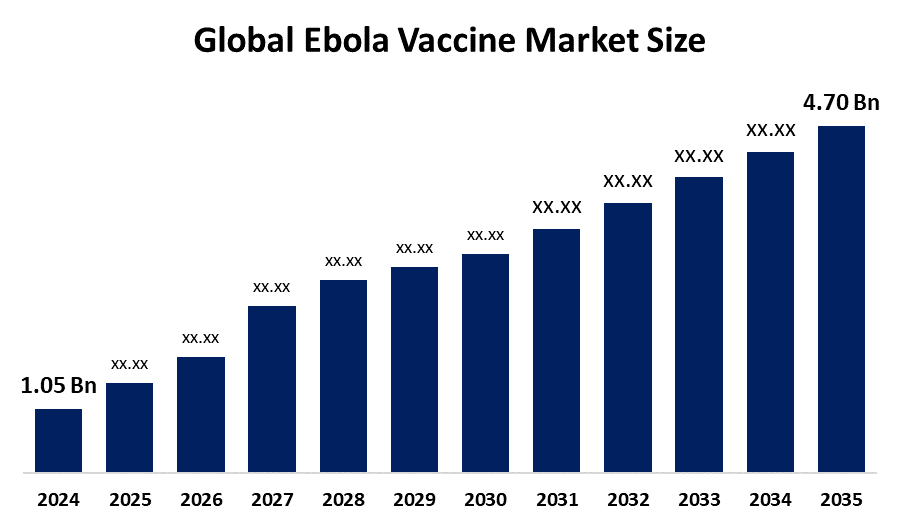
- The Global Ebola Vaccine Market Size Was Estimated at USD 1.05 Billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 14.60% from 2025 to 2035
- The Worldwide Ebola Vaccine Market Size is Expected to Reach USD 4.70 Billion by 2035
Get more details on this report -
Key Takeaways -
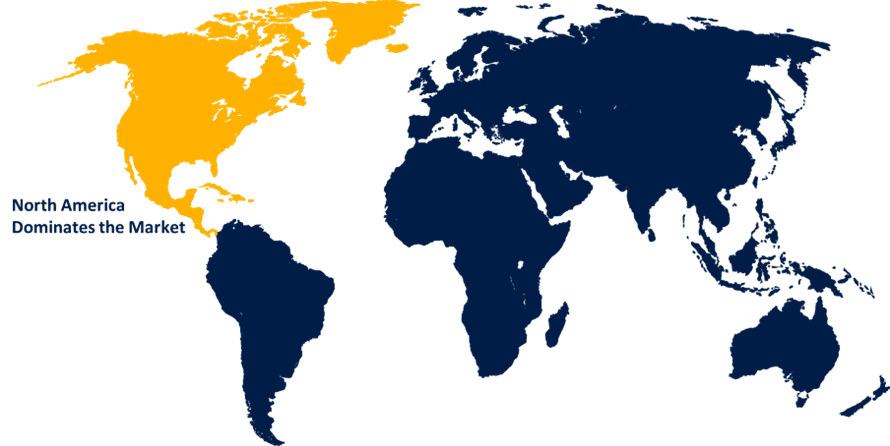
- North America dominated the market with the largest share in 2024.
- Asia Pacific is expected to grow the fastest during the forecast period.
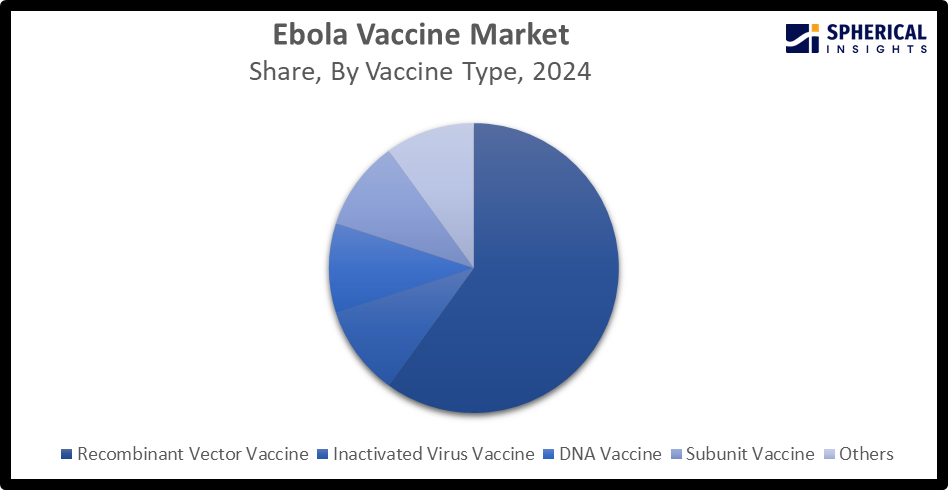
- By vaccine type, the recombinant vector vaccine segment accounted for the largest market share of about 62.0%, while the DNA vaccine is the emerging segment in the market.
- By end user, the government health departments segment is anticipated to hold the largest share, on the other hand, the research institutes segment is the fastest growing segment in the ebola vaccine market.
- By distribution channel, the direct sales segment is dominating the ebola vaccine market, while the online pharmacies segment is the fastest-growing channel.
According to a Research Report Published by Spherical Insights and Consulting, The Global Ebola Vaccine Market Size was Worth Around USD 1.05 Billion in 2024 and is Predicted to Grow to Around USD 4.70 Billion by 2035 with a Compound Annual Growth Rate (CAGR) of 14.60% from 2025 and 2035. The market for ebola vaccine has a number of opportunities to grow due to the expansion of preventative vaccination programs, especially in the high-risk African nations.
Key Trends in Medical Ebola Vaccine Market –
- Emphasis on rapid-response vaccines (single-dose)
- Advancement in viral vector technology
Market Overview
The Global Ebola Vaccine industry is the market involved in the development, production, stockpiling, and distribution of vaccines that protect against the Ebola virus (mainly used during outbreaks and for high-risk populations like healthcare workers). An Ebola vaccine is a vaccine used to prevent Ebola virus disease (Ebola), with notable candidates including rVSV-ZEBOV (Ervebo), which was approved by the US FDA in 2019. The vaccine uses a live, attenuated recombinant vesicular stomatitis virus (VSV) modified to express the Ebola virus glycoprotein.
Innovation and market expansion are anticipated as a result of major players' growing R&D expenditures and expanding partnerships. For instance, in January 2026, A vaccine used to help protect against Zaire ebolavirus, which is one of the world’s most severe infectious diseases could become more affordable and easier to deploy in low-resource settings thanks to a new collaboration between CEPI and MSD. Backed by up to $30 million in CEPI funding, MSD would leverage Hilleman Laboratories, a joint venture of MSD and Wellcome, to develop an Ebola vaccine with an updated manufacturing process that is designed to help make the vaccine more affordable and accessible for low- and middle-income countries.
Report Coverage
This research report categorizes the Global Ebola Vaccine Market Size based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the ebola vaccine market. Recent market developments and competitive strategies such as expansion, type launch, development, partnership, merger, and acquisition have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the ebola vaccine market.
Global Ebola Vaccine Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 1.05 Billion |
| Forecast Period: | 2024-2035 |
| Forecast Period CAGR 2024-2035 : | 14.60% |
| 2035 Value Projection: | USD 4.70 Billion |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 245 |
| Tables, Charts & Figures: | 100 |
| Segments covered: | By Region ,By Vaccine Type |
| Companies covered:: | Merck & Co., Johnson & Johnson, Bavarian Nordiac A/S, BioProtection Systems Corporation, Pfizer Inc., GlaxoSmithKline plc, Novavax, Inc., Serum Institute of India Pvt. Ltd., Sanofi Pasteur, Valneva SE, and Other Key Players |
| Pitfalls & Challenges: | COVID-19 Empact, Challenges, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
Government vaccination program
Two main vaccines, namely ERVEBO and Zabdeno/Mvabea, are used in government programs and global stockpiles to prevent Zaira ebolavirus. Furthermore, Gavi, the Vaccine Alliance launched a proactive 2024 program to immunize front-line workers in at-risk areas, supporting national responses.
WHO requires USD 21 million for supporting the Government of the Democratic Republic of the Congo to scale up response operations to interrupt all transmission chains, control the outbreak, and mitigate impact on human health, which would be achieved by implementing comprehensive integrated surveillance, alert management, testing, contact tracing, and safe clinical care as well as increasing participation in outbreak prevention and control.
Rising occurrence of Ebola outbreaks and the advancement in vaccine technology
The major Ebola outbreaks were estimated to have occurred in African nations, especially in Sudan, the DRC, Uganda, and Gabon. As per the WHO report, the average Ebola disease case fatality rate is around 50%, with a case fatality rate varying from 25-90% in past outbreaks. Innovation like recombinant viral vectors and mRNA vaccines that provide better efficacy and safety profiles, is driving the market growth of the Ebola vaccine.
Restraining Factors
Logistical hurdles and pricing pressure in low-income regions
Logistical hurdles, including extreme cold chain requirements, and accessibility of licensed vaccines for high-threat infectious diseases, are challenging factors in the Ebola vaccine market.
Market Segmentation
The Ebola Vaccine Market share is classified into vaccine type and end user.
- The recombinant vector vaccine segment dominated the market with the largest share of about 62.0% in 2024 and is projected to grow at a substantial CAGR during the forecast period.
Based on the vaccine type, the Global Ebola Vaccine Market Size is divided into recombinant vector vaccine, inactivated virus vaccine, DNA vaccine, subunit vaccine, and others. Among these, the recombinant vector vaccine segment dominated the market with the largest share of about 62.0% in 2024 and is projected to grow at a substantial CAGR during the forecast period. This is due to its superior efficacy and advanced technology applications. Recombinant vector Ebola vaccines are prepared using reverse genetics technology to create attenuated, modified viruses. While, the DNA vaccine involves designing plasma DNA constructs that encode specific, highly immunogenic Ebola virus proteins. The segment is the fastest-growing segment in the Ebola vaccine market, due to its scientific, manufacturing, and strategic advantages over traditional vaccine platforms.
Get more details on this report -
- The government health departments segment accounted for the largest market share of about 50.0% in 2024 and is projected to grow at a substantial CAGR during the forecast period.
Based on the end user, the Global Ebola Vaccine Market Size is divided into hospitals, government health departments, research institutes, NGOs, and others. Among these, the government health departments segment accounted for the largest market share of about 50.0% in 2024 and is projected to grow at a substantial CAGR during the forecast period. This is driven by the government funding, regulatory support, and emergency stockpiling. Further, the research institutes segment is the fastest-growing segment, driven by increasing partnerships between pharmaceutical companies and research institutes like the National Institutes of Health (NIH).
Regional Segment Analysis of the Ebola Vaccine Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the Ebola Vaccine market over the predicted timeframe.
North America is anticipated to hold the largest share of about 40.0% in the Global Ebola Vaccine Market Size over the predicted timeframe. The market for Ebola Vaccines has been driven by the region's increased R&D investment, government funding, and presence of key biopharma players. The U.S. is leading the North America Ebola Vaccine market, due to strong government investment, presence of key companies, advanced R&D infrastructure, and procurement & stockpiling.
Get more details on this report -
Asia Pacific is expected to grow at a rapid CAGR of approximately 9.5% in the Global Ebola Vaccine Market Size during the forecast period. The Asia Pacific area has a thriving market for Ebola Vaccine driven by an increasing awareness of infectious diseases and government support. Further, the major vaccine manufacturing and development sector in the region is responsible for market growth. China is holding the largest share within the Asia Pacific region, driven by strong government support, a growing biopharmaceutical industry, and preventive measures against emerging diseases.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the Global Ebola Vaccine Market Size, along with a comparative evaluation primarily based on their type of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes type development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Merck & Co.
- Johnson & Johnson
- Bavarian Nordiac A/S
- BioProtection Systems Corporation
- Pfizer Inc.
- GlaxoSmithKline plc
- Novavax, Inc.
- Serum Institute of India Pvt. Ltd.
- Sanofi Pasteur
- Valneva SE
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting And Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In January 2026, South Korean-based biotechnology company SK biosciences announced its involvement with the Coalition for Epidemic Preparedness Innovations (CEPI) and pharmaceutical company MSD. The collaboration would focus on improving formulations for a Zaire ebolavirus vaccine, with CEPI providing a $30 million grant toward research and innovation of the manufacturing process.
- In January 2026, Merck & Co., opens a new tab unit MSD and the Coalition for Epidemic Preparedness Innovations, they have partnered on a $30 million program to develop an updated version of the drugmaker's Ebola vaccine aimed at improving affordability and access for low- and middle-income countries.
- In February 2025, Uganda’s Ministry of Health, the World Health Organization (WHO) and other partners launched a first-ever clinical efficacy trial for a vaccine from Ebola from the Sudan species of the virus, and at an unprecedented speed for a randomized vaccine trial, in an emergency.
- In August 2023, the FDA approved an expanded indication for Ebola Zaire Vaccine, Live (Ervebo; Merck) to include individuals 12 months of age and older. The vaccine is a single-dose, intramuscular injection designed to prevent Ebola virus disease (EVD) caused by the Zaire Ebola virus.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the Global Ebola Vaccine Market Size based on the below-mentioned segments:
Global Ebola Vaccine Market, By Vaccine Type
- Recombinant Vector Vaccine
- Inactivated Virus Vaccine
- DNA Vaccine
- Subunit Vaccine
- Others
Global Ebola Vaccine Market, By End User
- Hospitals
- Government Health Departments
- Research Institutes
- NGOs
- Others
Global Ebola Vaccine Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
-
1. What is the market size of the Ebola Vaccine market?The global Ebola Vaccine market size is expected to grow from USD 1.05 Billion in 2024 to USD 4.70 Billion by 2035, at a CAGR of 14.60% during the forecast period 2025-2035.
-
2. Which region holds the largest share of the Ebola Vaccine market?North America is anticipated to hold the largest share of the Ebola Vaccine market over the predicted timeframe.
-
3. What is the forecasted CAGR of the Global Ebola Vaccine Market from 2024 to 2035?The market is expected to grow at a CAGR of around 14.60% during the period 2024–2035.
-
4. Who are the top companies that are involved in the Global Ebola Vaccine Market?Key players include Merck & Co., Johnson & Johnson, Bavarian Nordiac A/S, BioProtection Systems Corporation, Pfizer Inc., GlaxoSmithKline plc, Novavax, Inc., Serum Institute of India Pvt. Ltd., Sanofi Pasteur, and Valneva SE.
-
5. What are the main drivers in the Ebola Vaccine market?Government vaccination programs, rising occurrence of Ebola outbreaks, and the advancement in vaccine technology, are major market growth drivers of the ebola vaccine market.
-
6. What challenges are limiting the adoption of Ebola Vaccine?Factors like the Logistical hurdles and pricing pressure remain key restraints in the Ebola Vaccine market.
-
7. What are the key trends in the Ebola Vaccine market?The increasing emphasis on rapid-response vaccines (single-dose) and advancement in viral vector technology are major key trends in the Ebola Vaccine market.
Need help to buy this report?