Global Clinical Trial Equipment and Ancillary Solutions Market Size, Share, By Type (Diagnostic equipment, Laboratory instruments, and Ancillary solutions), By Service (Rental & leasing services, Supply and logistics services, Regulatory compliance services, and Software services), By End User (Pharmaceutical & biotechnology companies, Contract research organizations, Medical device companies, and Others), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2026 - 2035.
Industry: HealthcareGlobal Clinical Trial Equipment And Ancillary Solutions Market Insights and Forecasts to 2035
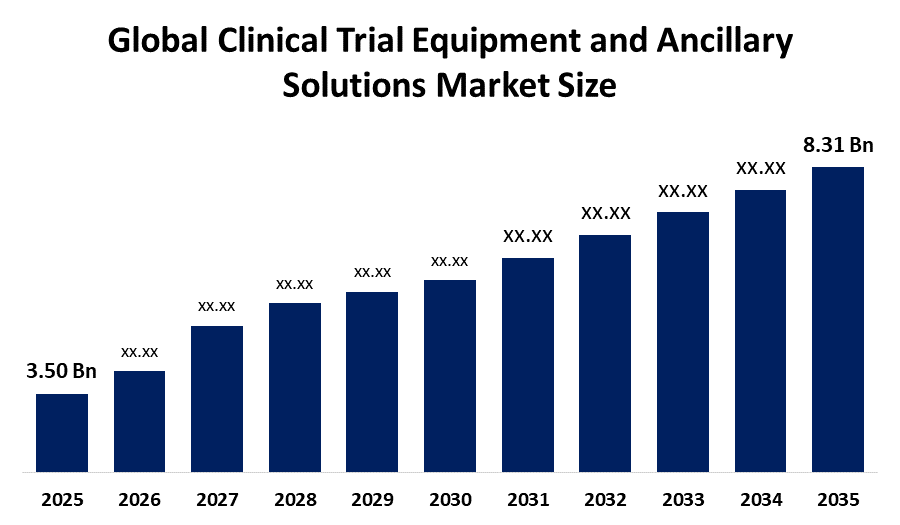
- The Global Clinical Trial Equipment And Ancillary Solutions Market Size Was Estimated at USD 3.50 billion in 2025
- The Market Size is Expected to Grow at a CAGR of around 9.03% from 2026 to 2035
- The Worldwide Clinical Trial Equipment And Ancillary Solutions Market Size is Expected to Reach USD 8.31 billion by 2035
- Asia Pacific is expected to grow the fastest during the forecast period.
Get more details on this report -
According to a research report published by Spherical Insights, The Global Clinical Trial Equipment and Ancillary Solutions Market Size was worth around USD 3.50 Billion in 2025 and is predicted to grow to around USD 8.31 Billion by 2035 with a compound annual growth rate (CAGR) of 9.03% from 2026 to 2035. The global clinical trial equipment and ancillary solutions market is driven by expanding biopharmaceutical pipelines, increasing demand for remote trial support, stringent quality standards, rising multi-site studies, and the need for efficient inventory and compliance management.
Market Overview
The global clinical trial equipment and ancillary solutions market refers to the industry that supplies medical devices, logistics, calibration, and support services required to conduct clinical trials efficiently and in compliance with protocols. The market is experiencing growth due to increasingly complex clinical trials, rising pharmaceutical and biotechnology research and development expenditures, and the growing demand for standardized, high-quality data collection methods. In May 2023, the FDA issued draft guidance on decentralized clinical trials, supporting remote monitoring, the use of digital health technologies, and home-based data collection. This regulatory direction has boosted demand for specialized portable equipment and direct-to-patient logistics tools used in decentralized and hybrid clinical trials. The market expansion is driven by government initiatives, including funding programs, regulatory reforms, and support for clinical research infrastructure development. In March 2025, Almac Group, through Almac Pharma Services, invested approximately USD 84 million in a new commercial manufacturing facility to expand oral dose production and enhance services supporting clinical trial supply and development operations. The market shows major trends, including the transition to decentralized, patient-centric trials, the rise of outsourcing to specialized service providers, and expansion into developing countries. The combination of AI-based data management, IoT-enabled equipment tracking, wearable devices, cloud platforms, and blockchain integration technologies provides real-time monitoring, supply chain visibility, and data accuracy, resulting in improved trial efficiency and outcomes.
Report Coverage
This research report categorizes the global clinical trial equipment and ancillary solutions market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the global clinical trial equipment and ancillary solutions market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyzes their core competencies in each sub-segment of the clinical trial equipment and ancillary solutions market.
Global Clinical Trial Equipment and Ancillary Solutions Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2025 |
| Market Size in 2025: | USD 3.50 Billion |
| Forecast Period: | 2026 – 2035 |
| Forecast Period CAGR 2026 – 2035 : | CAGR of 9.03% |
| 026 – 2035 Value Projection: | USD 8.31 Billion |
| Historical Data for: | 2020-2024 |
| No. of Pages: | 240 |
| Tables, Charts & Figures: | 95 |
| Segments covered: | By Type By Service |
| Companies covered:: | Thermo Fisher Scientific Inc, Parexel International Corporation, ICON plc, IQVIA Holdings Inc, Veeva Systems Inc, Oracle Corporation, Medidata Solutions, Ancillare, LP, Imperial Clinical Research Services, Woodley Equipment Company Ltd, Quipment SAS, IRM, Marken, Myonex, Eurofins Scientific SE, Others, and Key Players |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
The global market for clinical trial equipment and ancillary solutions experiences growth because of rising clinical trials, which require more intricate equipment solutions across various medical fields, especially in oncology and rare disease research. The increasing demand for specialized equipment and support services results from pharmaceutical and biotechnology companies increasing their research and development expenditures while they search for better methods to manage their trial operations. The demand for logistical solutions, remote monitoring systems, and standardized equipment has increased because of the trend toward decentralized and hybrid trial methods. The practice of outsourcing to contract research organizations, the international expansion of clinical trials, and the enforcement of stringent regulatory standards drive organizations to implement dependable additional solutions that maintain data precision and regulatory adherence and enable fast trial processes.
Restraining Factors
The global clinical trial equipment and ancillary solutions market faces restraints because equipment sourcing, maintenance, and worldwide distribution require high operational and logistics expenses. The complex regulatory requirements that exist in different regions create two problems for companies because they slow down the approval process while increasing their compliance requirements. Multi-site trials face operational challenges because they lack standardized procedures and fail to achieve effective coordination between different testing sites. The equipment malfunction risks, data inaccuracy risks, and supply chain disruption risks all create dangers that can harm both trial schedules and overall results.
Market Segmentation
The global clinical trial equipment and ancillary solutions market share is classified into type, service, and, end user.
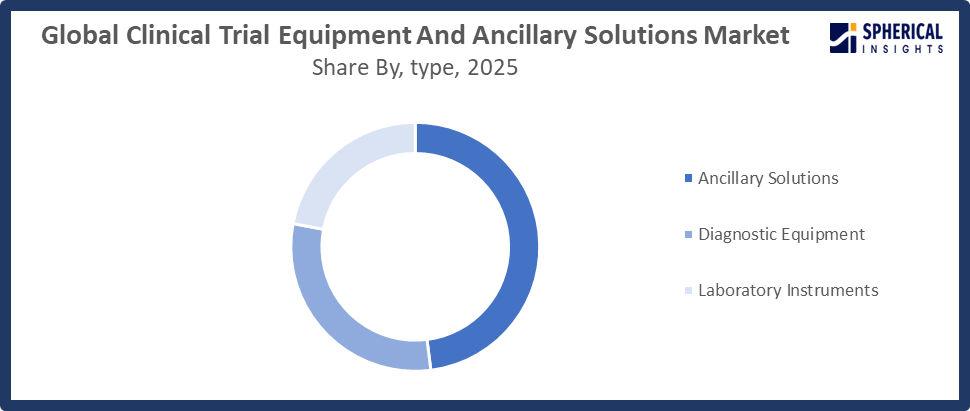
- The ancillary solutions segment accounted for the largest share in 2025, approximately 48%, and is anticipated to grow at a significant CAGR during the forecast period.
Based on the type, the global clinical trial equipment and ancillary solutions market is divided into diagnostic equipment, laboratory instruments, and ancillary solutions. Among these, the ancillary solutions segment accounted for the largest share in 2025, approximately 48%, and is anticipated to grow at a significant CAGR during the forecast period. This is because it provides comprehensive support that goes beyond equipment, including supply chain management, temperature-controlled logistics, patient kits, and trial monitoring services. Rising adoption of decentralized and hybrid trials, increasing trial complexity, and the need for accurate, timely data drive demand. Outsourcing to specialized service providers further boosts reliance on these solutions.
Get more details on this report -
- The supply and logistics services segment accounted for the highest market revenue in 2025, approximately 42%, and is anticipated to grow at a significant CAGR during the forecast period.
Based on the service, the global clinical trial equipment and ancillary solutions market is divided into rental & leasing services, supply and logistics services, regulatory compliance services, and software services. Among these, the supply and logistics services segment accounted for the highest market revenue in 2025, approximately 42% and is anticipated to grow at a significant CAGR during the forecast period. This is because efficient supply chain solutions, including temperature-controlled transport and inventory management, are essential for timely delivery of equipment and materials, ensuring compliance and reliability in increasingly complex and global clinical trials.
- The pharmaceutical & biotechnology companies segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the end user, the global clinical trial equipment and ancillary solutions market is divided into the pharmaceutical & biotechnology companies, contract research organizations, medical device companies, and others. Among these, the pharmaceutical & biotechnology companies segment dominated the market in 2025, approximately 50% and is projected to grow at a substantial CAGR during the forecast period. The dominance is driven by increasing R&D investments, a rising number of clinical trials, demand for advanced trial management solutions, and the need for reliable equipment and ancillary services to ensure data accuracy, compliance, and timely completion of complex and large-scale trials.
Regional Segment Analysis of the Global Clinical Trial Equipment And Ancillary Solutions Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Get more details on this report -
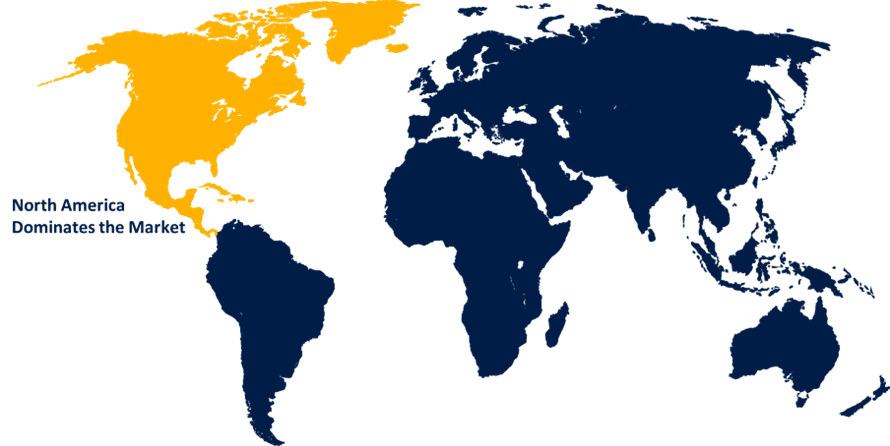
North America is anticipated to hold the largest share of the global clinical trial equipment and ancillary solutions market over the predicted timeframe.
North America is anticipated to hold the largest share of the global clinical trial equipment and ancillary solutions market over the predicted timeframe, approximately 40% market share. The growth is driven by the concentration of major pharmaceutical and biotechnology companies, well-developed clinical research infrastructure, substantial R&D investments, and supportive regulatory frameworks. These factors enable efficient planning and execution of large-scale clinical trials, ensure compliance with international standards, and promote adoption of advanced equipment and ancillary solutions, strengthening leadership in the global market.
The Asia Pacific region is expected to grow at the fastest rate during the forecast period, approximately 28% market share. The market growth is fueled by increasing outsourcing of clinical trials, expanding healthcare infrastructure, rising number of contract research organizations, favorable government initiatives, and growing demand for cost-effective, efficient trial solutions, driving rapid adoption of clinical trial equipment and ancillary services across emerging economies.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global clinical trial equipment and ancillary solutions market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Thermo Fisher Scientific Inc
- Parexel International Corporation
- ICON plc
- IQVIA Holdings Inc
- Veeva Systems Inc
- Oracle Corporation
- Medidata Solutions
- Ancillare, LP
- Imperial Clinical Research Services
- Woodley Equipment Company Ltd
- Quipment SAS
- IRM
- Marken
- Myonex
- Eurofins Scientific SE
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In January 2025, Ancillare announced the launch of its enhanced Cold Chain Management service for clinical trial supplies, aimed at maintaining temperature integrity, expanding global logistics capabilities, and providing real-time tracking and visibility to ensure safe handling and delivery across clinical trial sites.
- In February 2024, FedEx Express inaugurated the FedEx Life Science Center (LSC) in Mumbai, India, to support clinical trial storage and logistics needs for healthcare clients both in India and internationally. The facility is part of FedEx’s global network of Life Science Centers in Japan, South Korea, Singapore, the USA, and the Netherlands, strengthening its end-to-end supply chain capabilities for temperature-sensitive clinical trial materials and pharmaceutical products.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the global clinical trial equipment and ancillary solutions market based on the below-mentioned segments:
Global Clinical Trial Equipment and Ancillary Solutions Market, By Type
- Diagnostic equipment
- Laboratory instruments
- Ancillary solutions
Global Clinical Trial Equipment and Ancillary Solutions Market, By Service
- Rental & leasing services
- Supply and logistics services
- Regulatory compliance services
- Software services
Global Clinical Trial Equipment and Ancillary Solutions Market, By End User
- Pharmaceutical & biotechnology companies
- Contract research organizations
- Medical device companies
- Others
Global Clinical Trial Equipment and Ancillary Solutions Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
-
1. What does the clinical trial equipment and ancillary solutions market include?A: It covers medical devices, laboratory instruments, patient monitoring tools, temperature-controlled storage, shipping containers, software platforms, and services such as logistics, regulatory compliance, and trial support.
-
2. Why is this market growing rapidly?A: The market grows due to increasing clinical trials, adoption of decentralized and hybrid trial designs, demand for accurate and standardized data, and rising R&D investments by pharmaceutical and biotechnology companies.
-
3. How do these solutions improve clinical trials?A: They enhance operational efficiency, maintain sample integrity, ensure compliance with global regulations, reduce delays, and provide real-time data tracking for better trial outcomes.
-
4. What role does technology play in this market?A: Technologies such as IoT-enabled monitoring, AI-based analytics, cloud platforms, and blockchain improve supply chain management, patient engagement, and data accuracy.
-
5. What are the main challenges faced by the market?A: High operational costs, complex global regulations, logistics hurdles, equipment maintenance, and integrating new technologies into existing trial workflows.
-
6. Which regions show the most potential for growth?A: While North America and Europe dominate due to established infrastructure, Asia Pacific is the fastest-growing region driven by expanding clinical trials, emerging CROs, and healthcare infrastructure development.
Need help to buy this report?