China Pharmacovigilance Market Size, Share, and COVID-19 Impact Analysis, By Type (Intensified ADR Reporting, Target Spontaneous ARD Reporting, Cohort Event Monitoring, HER Mining), By End User (Pharmaceutical Companies, Biotechnology Companies, Contract Research Organizations CROs, Hospitals, and Healthcare Providers), China Pharmacovigilance Market Insights, Industry Trend, Forecasts to 2035
Industry: HealthcareChina Pharmacovigilance Market Size Insights Forecasts to 2035
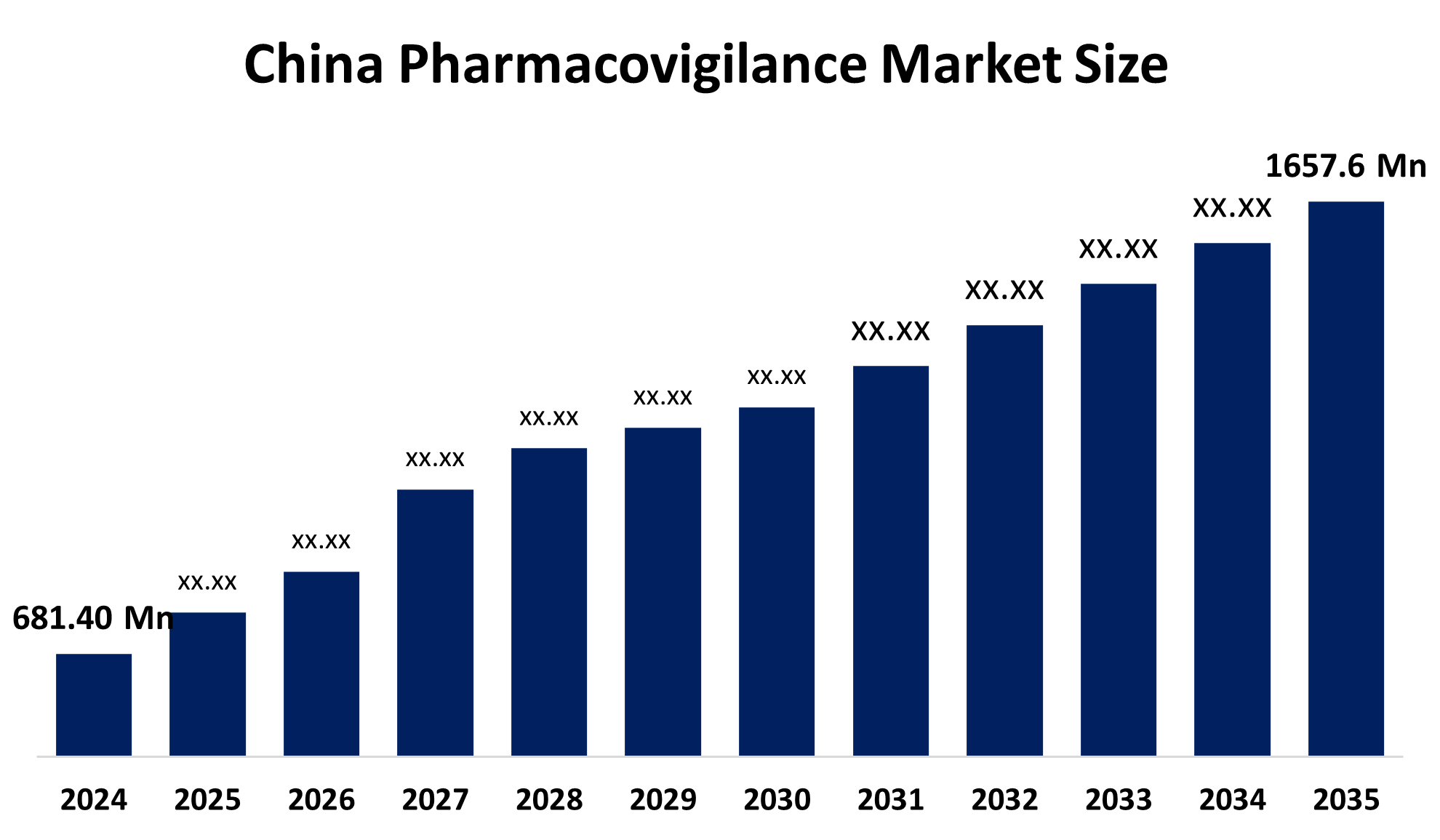
- The China Pharmacovigilance Market Size Was Estimated at USD 681.40 Million in 2024
- The Market Size is Expected to Grow at a CAGR of Around 8.42% from 2025 to 2035
- The China Pharmacovigilance Market Size is Expected to Reach USD 1657.6 Million by 2035
Get more details on this report -
According to a research report published by Spherical Insights & Consulting, China Pharmacovigilance Market Size is Anticipated to Reach USD 1657.6 Million by 2035, Growing at a CAGR of 8.42% from 2025 to 2035. The market is driven by the national medical products administration NMPA requiring marketing authorization holders to take responsibility for drug safety, and rapid digitalization and a increasing outsourcing, and advanced AI-driven technology integration.
Market Overview
The China Pharmacovigilance Market refers to a specialized regulatory framework for detection, assessment, understanding, and prevention of adverse drug reactions ADRs and other medicine-related problems throughout the entire product lifestyle. Its experiencing rapid growth driven by stringent NMPA regulations, increased drug innovations. and the pharmaceutical companies are increasingly outsourcing PV activities to contract research organizations CROs to enhance efficiency and ensure compliance. The key trends are also including AI-driven safety analytics, cloud-based platform adoption, and a focus on phase IV post-marketing studies to manage adverse drug reactions ADRs.
In NMPA requires all holders to establish a PV system with a local responsible person RPPV and submit periodic safety update reports PSURs and individual case safety reports ICSRs. The key activates are mandatory such as electronic submission of individual case safety reports ICSR, periodic safety update reports PSUR, continuous signal detection, and maintenance of a pharmacovigilance system master file PSMF.
The innovations in China Pharmacovigilance Market such as the AI and machine learning companies are deploying AI for big data analytics, enhancing predictive capabilities for risk management. the digital integration with electronic health records EHRs enables seamless, real-time data exchange for faster signal detection. The local technology enables CROs are utilizing AI-based platforms for clinical trial documents handling, improving efficiency and reducing costs.
Report Coverage
This research report categorizes the market for the China Pharmacovigilance Market based on various segments and regions, and forecasts revenue growth and analyses trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the China pharmacovigilance market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the China pharmacovigilance market.
China Pharmacovigilance Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 681.40 Million |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | 8.42% |
| 2035 Value Projection: | USD 1657.6 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 225 |
| Tables, Charts & Figures: | 102 |
| Segments covered: | By Type, By End User |
| Companies covered:: | IQVIA (clinical data and PV services), Parexel international corporation, ICON plc, Accenture, Cognizant, Oracle health sciences (PV software), and Other Key Players |
| Pitfalls & Challenges: | and COVID-19 Impact Analysis |
Get more details on this report -
Driving Factors
The China Pharmacovigilance Market is driven by strict regulatory requirements in NMPA, high-volume drug innovation, and a shift toward digital safety monitoring. In drug and biotech innovations the China is a global leader in drug developments, with 46% of new molecular entities entering trails,creating massive demand for pre-and post- marketing safety monitoring. The digital transformation and AI they need to process vast volumes of the ADR data has driven AI, machine learning, and electronic health record EHR mining, which are key to efficient signal detection. The rising healthcare expenditure they are increasing disposable income and expanding healthcare infrastructure are driving higher drug consumption, which necessitates robust, comprehensive safety systems.
Restraining Factors
China Pharmacovigilance Market faces restraints such as data quality and infrastructure reliance on spontaneous reporting limits data quality, necessitating more active, technology-driven surveillance, which requires expensive infrastructure. and high initial investment for PV software and operational costs, coupled with, rising labour expenses, pose barriers, particularly for small and mid-sized firms.
Market Segmentation
The China pharmacovigilance market share is categorized type and by end user.
- The target spontaneous ARD reporting segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period.
The China Pharmacovigilance Market is segmented by type Intensified ADR reporting, target spontaneous ARD reporting, cohort event monitoring, HER mining. Among these, The Target spontaneous ARD reporting segment accounted for the largest revenue market share in 2024 and is expected to grow at a significant CAGR during the forecast period. In the targeted spontaneous reporting TSR is increasingly integrated within hospital networks and post-marketing projects, focusing on high-risk, newly listed, and orphan drug to overcome underreporting. In according to NMPA announcement no.66 in 2018, MAHs must report any suspected adverse drug reactions ADRs immediately via the China adverse drug reaction monitoring system CADRMS. It concentrates on specific drugs known for high risk, such as antiretrovirals, new drugs rather than all medications.
- The pharmaceutical companies segment dominated the market in 2024 and is projected to grow at a substantial CAGR during the forecast period.
The China Pharmacovigilance Market is segmented by end user into pharmaceutical companies, biotechnology companies, contract research organizations CROs, hospitals and healthcare providers. Among these, the pharmaceutical companies segment dominated the market in 2024 and is projected to grow at a substantial CAGR during the forecast period. the pharmaceutical companies dominated market. Because of mandatory drug safety monitoring, high-risk product development, and the need for compliance. The product lifestyle management increased focus on phase IV trails of post-marketing surveillance for monitoring drug efficacy and safety on a large scale. In 2026 revisions to the drug administration law, the MAH is strictly accountable for the entire life cycle of drug, including pre-clinical, and post-market safety. In the high-risk products portfolios such as the rise of oncology, biopharmaceuticals, and complex biologics in China demands intensive safety monitoring, forcing pharma companies to invest in or contract advanced PV systems.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the China Pharmacovigilance Market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborate analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- IQVIA (clinical data and PV services)
- Parexel international corporation
- ICON plc
- Accenture
- Cognizant
- Oracle health sciences (PV software)
- Other
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the China, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the China Pharmacovigilance Market based on the below-mentioned segments.
China pharmacovigilance market, By Type
- Intensified ADR Reporting
- Target Spontaneous ARD Reporting
- Cohort Event Monitoring
- HER Mining
China pharmacovigilance market, By End user
- Pharmaceutical Companies
- Biotechnology Companies
- Contract Research Organizations CROs
- Hospitals and Healthcare Providers
- Others
Frequently Asked Questions (FAQ)
-
Q: What is the China pharmacovigilance market size?China pharmacovigilance market size is expected to grow from USD 681.40 million in 2024 to USD 1657.6 million by 2035, growing at a CAGR of 8.42% during the forecast period 2025-2035.
-
Q: What are the key growth drivers of the market?Market growth is driven by strict regulatory requirements in NMPA, high-volume drug innovation, and a shift toward digital safety monitoring.
-
Q: What factors restrain the China pharmacovigilance market?The China pharmacovigilance market faces restraints such as data quality and infrastructure reliance on spontaneous reporting limits data quality, necessitating more active, technology-driven surveillance, which requires expensive infrastructure
-
Q: How is the market segmented by type?The market is segmented into intensified ADR reporting, target spontaneous ARD reporting, cohort event monitoring, HER mining
-
Q: Who are the key players in the China pharmacovigilance market?Key companies include IQVIA (clinical data and PV services), Parexel international corporation, ICON plc, Accenture, Cognizant, Oracle health sciences (PV software)
Need help to buy this report?