Global Blood Grouping Reagents Market Size, Share, By Product (Consumables, Instruments, and Service & Software), By Test Type (Blood Group & Phenotype, Antibody Screening, Cross-Matching Tests, Antibody Identification, Coombs Tests, and Antigen Typing), By Technique (PCR-Based & Microarray Techniques, Assay-Based Techniques, Massively Parallel Sequencing Techniques, Serology, and Others), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2026-2035
Industry: HealthcareGlobal Blood Grouping Reagents Market Insights Forecasts to 2035
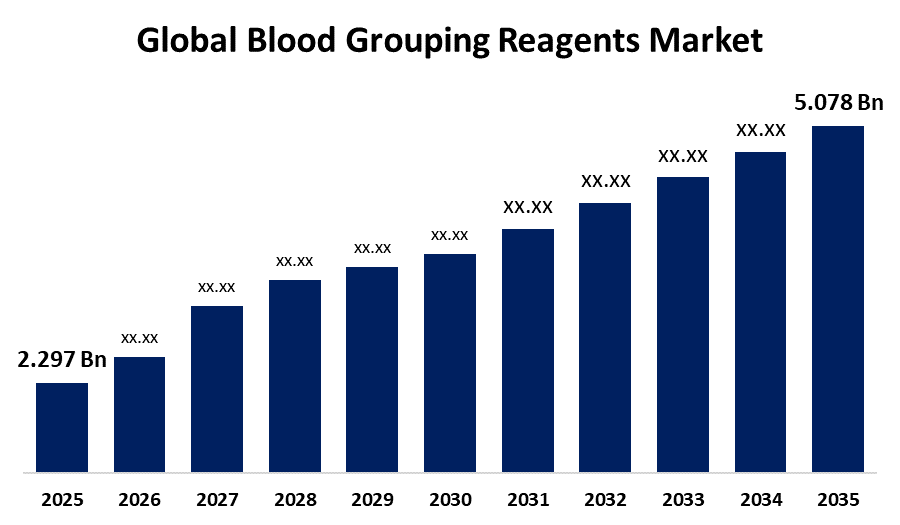
- The Global Blood Grouping Reagents Market Size Was Estimated at USD 2.297 Billion in 2025
- The Market Size is Expected to Grow at a CAGR of around 8.26% from 2026 to 2035
- The Worldwide Blood Grouping Reagents Market Size is Expected to Reach USD 5.078 Billion by 2035
- Asia Pacific is expected to grow the fastest during the forecast period.
Get more details on this report -
According to a research report published by Spherical Insights and Consulting, the global blood grouping reagents market size was worth around USD 2.297 billion in 2025 and is predicted to grow to around USD 5.078 billion by 2035 with a compound annual growth rate (CAGR) of 8.26% from 2026 to 2035. Global expansion of automated analyzers, gel card systems, molecular genotyping, and AI enhanced point-of-care testing, innovations in high-specificity monoclonal reagents, digital lab integration, high-throughput platforms, and extended antigen profiling are all driving opportunities in the blood grouping reagents market.
Market Overview
The blood grouping reagents market refers to the global commercial market for chemical and biological substances used to determine blood types and compatibility for transfusions, organ transplants, and prenatal screening. The market includes reagents that consist of antisera, monoclonal antibodies, and molecular probes which scientists use for serological testing and genotyping analysis together with their associated automated systems, consumable materials, and diagnostic testing equipment. The market creates its main opportunities from technological advancements which include automated blood typing analyzers, high throughput screening systems, molecular genotyping technology, and point of care testing solutions because these innovations enhance testing precision while decreasing laboratory processing durations and enabling remote medical service provision.
Governments establish regulatory frameworks and standards for reagent quality and safety which government agencies use to control blood safety programs and public healthcare initiatives use to promote advanced diagnostic technologies through their funding programs. Supportive policies aimed at improving blood donation systems and enhancing laboratory capabilities also help drive market growth. For instance, in August 2025, the WHO reports that India collected 14.6 million blood units during the 2024-2025 period while total global blood collection reached 118.5 million units each year. The process of blood typing and cross-matching for each donated blood unit requires the use of ABO and Rh reagents which results in continuous and substantial needs for blood grouping reagents.
In February 2024, The U.S. CDC reported approximately 7 million blood donors every year while more than 14 million blood transfusions take place each year. The U.S. healthcare system requires approximately 29000 daily red blood cell units together with additional thousands of platelets and plasma units which directly increase the need for blood grouping reagents.
Report Coverage
This research report categorizes the blood grouping reagents market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the blood grouping reagents market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the blood grouping reagents market.
Global Blood Grouping Reagents Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2025 |
| Market Size in 2025: | USD 2.297 Billion |
| Forecast Period: | 2026-2035 |
| Forecast Period CAGR 2026-2035 : | 8.26% |
| 2035 Value Projection: | USD 5.078 Billion |
| Historical Data for: | 2020-2024 |
| No. of Pages: | 240 |
| Tables, Charts & Figures: | 112 |
| Segments covered: | By Product, By Test Type, By Technique and By Region |
| Companies covered:: | Bio-Rad Laboratories, Inc., Grifols, S.A., QuidelOrtho Corporation, Danaher Corporation, Immucor, Inc., Thermo Fisher Scientific Inc., Merck KGaA, DIAGAST, AliveDx, BAG Diagnostics Pvt. Ltd., Lorne Laboratories Limited, ARKRAY, Inc., Atlas Medical GmbH, Hemo bioscience, Inc., Others |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
Rapid technological innovation is one of the main drivers of the blood grouping reagents market's growth. Rising need for safe blood transfusions, increasing number of chronic diseases, and the growing understanding of blood compatibility testing, which is the main factor driving the market for blood grouping reagents. Innovation and investment in this field are driven by the growing use of automated analyzers, gel card systems, molecular genotyping, AI-assisted point-of-care testing, high-throughput screening platforms, and extended antigen profiling. Government initiatives that support blood safety together with regulatory backing and funding for new diagnostic equipment, help drive market expansion. Additionally the market demand strengthens as the number of hospitals, blood banks, and diagnostic centers increases together with the rising popularity of personalized transfusion therapy.
In June 2025, WHO’s World Blood Donor Day with the theme Give blood, give hope, together we save lives which aimed to raise public awareness about blood donation. The requirement for blood testing to conduct ABO and Rh typing creates a direct need for blood grouping reagents as organizations need to perform more tests when blood donations increase.
Restraining Factors
High development and deployment costs, Complex regulatory requirements, stringent quality standards, lengthy approval processes, supply chain disruption, low awareness in rural areas, competition from alternative diagnostic methods, and concerns over reagent stability and shelf life are the main factors restricting the blood grouping reagents market.
Market Segmentation
The blood grouping reagents market share is classified into product, test type, and technique.
- The consumables segment dominated the market in 2025, approximately 47%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the product, the blood grouping reagents market is divided into consumables, instruments, and service & software. Among these, the consumables segment dominated the market in 2025, approximately 47%, and is projected to grow at a substantial CAGR during the forecast period. The growing need for every blood typing and compatibility test, single-use and require frequent replenishment, and widespread use across hospitals, blood banks, and diagnostic labs ensuring steady consumption is driving the consumables industry.
- The blood group & phenotype segment accounted for the largest share in 2025, approximately 42%, and is anticipated to grow at a significant CAGR during the forecast period.
Based on the test type, the blood grouping reagents market is divided into blood group & phenotype, antibody screening, cross matching tests, antibody identification, coombs tests, and antigen typing. Among these, the blood group & phenotype segment accounted for the largest share in 2025, approximately 42%, and is anticipated to grow at a significant CAGR during the forecast period. Fundamental test type for determining a person’s ABO and Rh blood type, performed routinely in hospitals, blood banks, and prenatal screenings, creates high and consistent demand is driving the blood group & phenotype industry.
Get more details on this report -
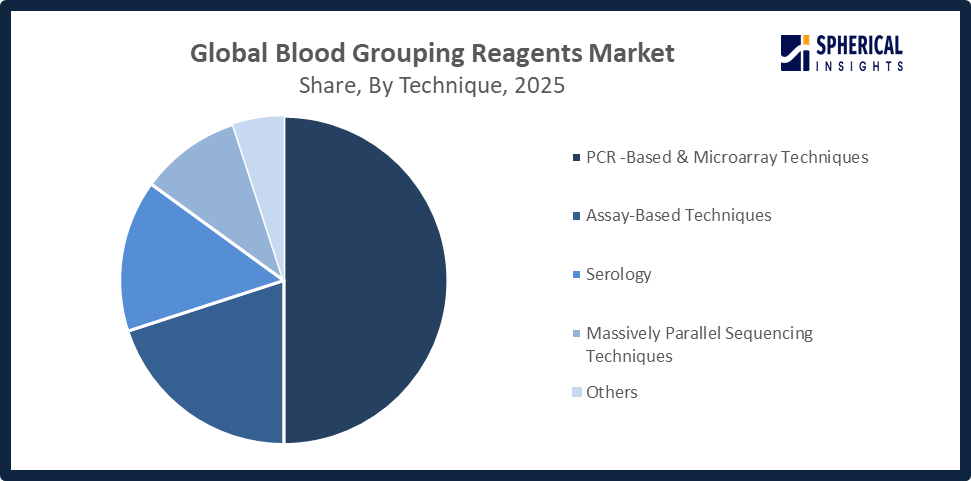
- The PCR-based & microarray techniques segment accounted for the highest market revenue in 2025, approximately 50%, and is anticipated to grow at a significant CAGR during the forecast period.
Based on the technique, the blood grouping reagents market is divided into PCR-based & microarray techniques, assay based techniques, massively parallel sequencing techniques, serology, and others. Among these, the PCR-based & microarray techniques segment accounted for the highest market revenue in 2025, approximately 50%, and is anticipated to grow at a significant CAGR during the forecast period. offer high accuracy, sensitivity, and the ability to detect rare or complex blood group antigens, growing adoption in advanced hospitals, reference labs, and personalized transfusion medicine, and rising demand for automated and high-throughput testing is bolstering PCR-based & microarray techniques market.
Regional Segment Analysis of the Blood Grouping Reagents Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
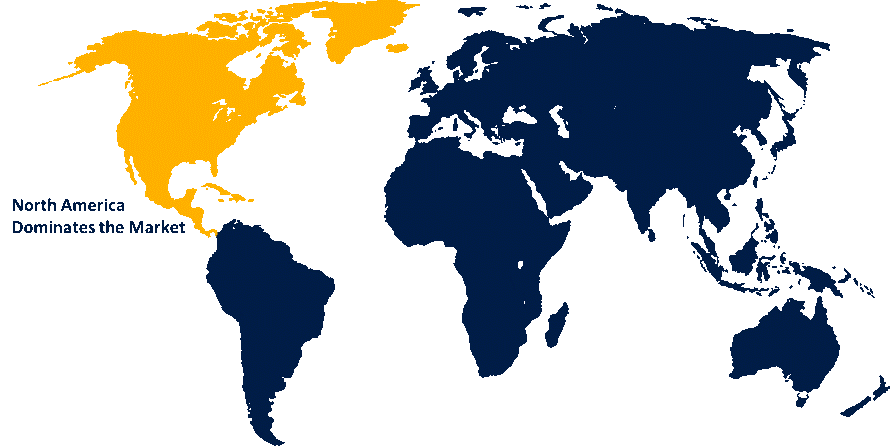
North America is anticipated to hold the largest share of the blood grouping reagents market over the predicted timeframe.
Get more details on this report -
North America is anticipated to hold the largest share of the blood grouping reagents market over the predicted timeframe. The regional leadership functions because the area shows advanced healthcare facilities, spends substantial amounts on healthcare services and hospitals, and blood banks use high-level diagnostic equipment. The combination of strong regulatory frameworks and transfusion safety requirements leads to hospitals adopting automated blood typing systems and molecular blood typing technology. The healthcare system needs to conduct regular tests because people donate blood at high rates and hospitals perform many surgical procedures and chronic diseases are on the rise. North America maintains its market leadership due to major reagent manufacturers and their ongoing research and development activities.
Government initiatives include the U.S. FDA controls blood and blood products together with licensed blood typing reagents to maintain their safe and effective use, October 2025 and CDC oversees blood safety and testing methods used across the U.S. creating need for regular ABO/Rh testing, February 2024, and Health Canada’s Blood Regulations mandate the use of approved test kits and reagents for blood grouping and compatibility testing, enforced by provincial blood operators.
Asia Pacific is expected to grow at a rapid CAGR in the blood grouping reagents market during the forecast period. China and India and Japan have emerged as major investors to support the expansion of automated and molecular blood typing technologies used in urban hospitals and diagnostic laboratories. The healthcare system experience rapid growth because healthcare facilities expand, funding increases, and people learn about safe blood transfusion methods. The need for advanced blood grouping reagents has increased because of the rising patient numbers, higher trauma case, and chronic disease rates that need blood compatibility tests and governments are launching programs to enhance blood donation systems and diagnostic capabilities in Asia Pacific.
Government launches include India’s MHFW and NBTC, December 2025, plans to improve blood safety and ensuring that all donated blood is screened and typed before use, sustaining demand for blood grouping and screening tests, China’s national law mandates testing and safety standards for all donated blood, overseen by the National Health Commission, June 2025, and Japan’s Red Cross and government run a national program ensuring all donated blood is typed and screened before use.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the blood grouping reagents market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Bio-Rad Laboratories, Inc.
- Grifols, S.A.
- QuidelOrtho Corporation
- Danaher Corporation
- Immucor, Inc.
- Thermo Fisher Scientific Inc.
- Merck KGaA
- DIAGAST
- AliveDx
- BAG Diagnostics Pvt. Ltd.
- Lorne Laboratories Limited
- ARKRAY, Inc.
- Atlas Medical GmbH
- Hemo bioscience, Inc.
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
Recent Development
- In June 2025, Devyser Diagnostics announced the global launch of Devyser Genomic Blood Typing, a next generation sequencing based solution that enables comprehensive genetic blood typing covering multiple antigen systems in a single streamlined test. This development advances molecular blood grouping capabilities beyond traditional serology methods.
- In April 2025, the U.S. Food and Drug Administration granted approval for Bio Rad Laboratories new line of rapid blood grouping reagents using microarray technology, marking a significant regulatory milestone that enables commercialization and broader clinical adoption of these advanced reagents.
- In March 2024, Grifols entered a strategic collaboration with the University of California, San Diego to co-develop next generation blood grouping reagents, combining Grifols manufacturing expertise with advanced immunology research.
- In January 2024, Thermo Fisher Scientific announced the launch of its RevealPLUS blood grouping reagents, designed to deliver improved accuracy and faster results in routine blood typing diagnostics to meet growing demand for reliable and rapid testing
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the blood grouping reagents market based on the below-mentioned segments:
Global Blood Grouping Reagents Market, By Product
- Consumables
- Instruments
- Service & Software
Global Blood Grouping Reagents Market, By Test Type
- Blood Group & Phenotype
- Antibody Screening
- Cross Matching Tests
- Antibody Identification
- Coombs Tests
- Antigen Typing
Global Blood Grouping Reagents Market, By Technique
- PCR-Based & Microarray Techniques
- Assay-Based Techniques
- Massively Parallel Sequencing Techniques
- Serology
- Others
Global Blood Grouping Reagents Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
-
1. What technologies do blood grouping reagents support?Blood grouping reagents are used in serological testing, molecular blood typing, and immunohematology assays. They support technologies such as ABO/Rh typing, extended antigen phenotyping, gel card testing, microplate assays, and automated blood typing analyzers for accurate and rapid results.
-
2. What types of blood samples are tested with these reagents?Blood grouping reagents are used on whole blood, plasma, or red blood cells collected from donors and patients. Testing may include manual tube methods, gel cards, and automated systems to determine ABO and Rh blood groups and other clinically relevant antigens.
-
3. What is the role of high-throughput and automated testing?High-throughput and automated testing systems allow laboratories and blood banks to process large volumes of samples efficiently, reduce human error, and maintain consistent results. Automation is critical for large-scale blood donation programs and hospital transfusion services.
-
4. What is molecular blood typing?Molecular blood typing uses genetic testing techniques to detect blood group antigens at the DNA level. This method complements traditional serology by identifying rare blood types, weak antigens, or complex phenotypes, improving transfusion safety.
-
5. What industries benefit from blood grouping reagents?Healthcare, hospital transfusion services, blood banks, diagnostic laboratories, and research institutions rely on blood grouping reagents for safe transfusions, donor screening, and clinical studies.
-
6. What is the expected market growth timeline?The blood grouping reagents market is expected to grow steadily through the 2020s, driven by increasing blood donation programs, government regulations, and technological advancements in automated and molecular testing systems. Market expansion is projected to continue through 2026–2030 with growing adoption globally.
Need help to buy this report?