Global Autologous Cell Therapy Market Size, Share, and COVID-19 Impact Analysis, By Therapy Type (Stem Cell Therapies, Non-Stem Cell Therapies, and Others), By Therapeutic Area (Oncology, Cardiovascular Disease (CVD), Musculoskeletal Disorders, Dermatology, and Others), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 - 2035.
Industry: HealthcareGlobal Autologous Cell Therapy Market Insights Forecasts to 2035
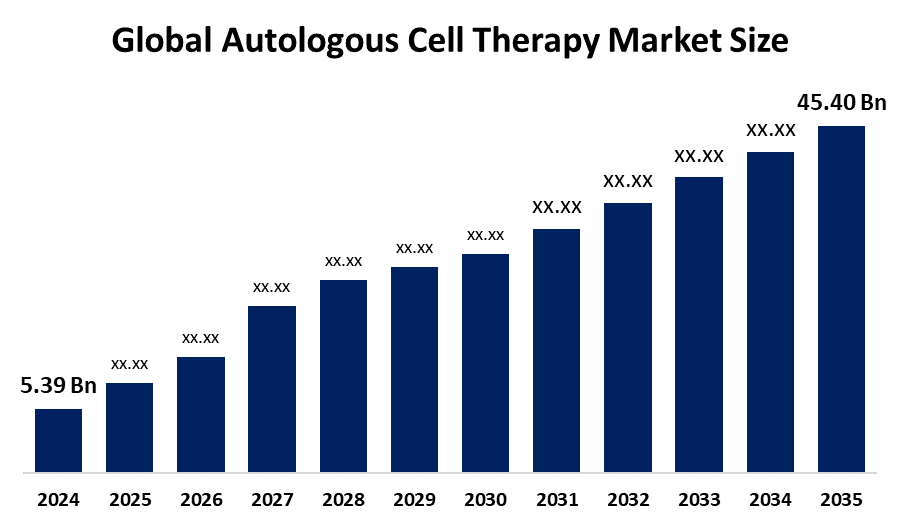
- The Global Autologous Cell Therapy Market Size Was Estimated at USD 5.39 Billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 21.38% from 2025 to 2035
- The Worldwide Autologous Cell Therapy Market Size is Expected to Reach USD 45.40 Billion by 2035
Get more details on this report -
Key Takeaways -
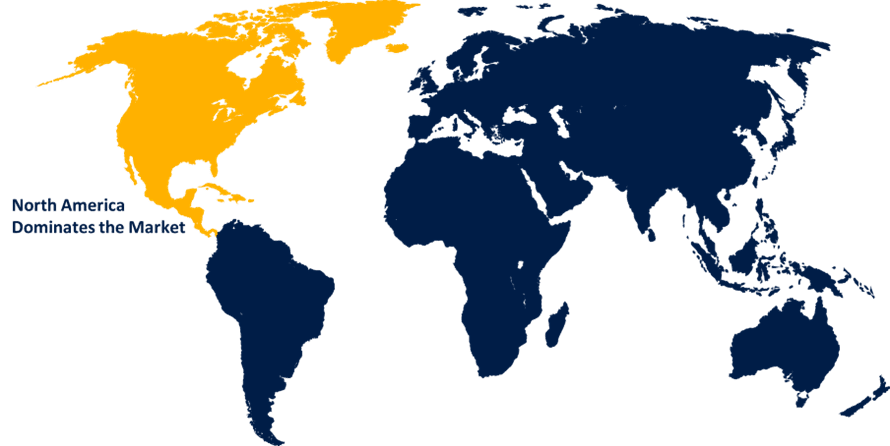
- North America dominated the market with the largest share in 2024.
- Asia Pacific is expected to grow the fastest during the forecast period.
- By therapy type, the non-stem cell therapies segment dominated the market with about 90.0% share in 2024, while the stem cell therapies segment is growing significantly during the forecast period.
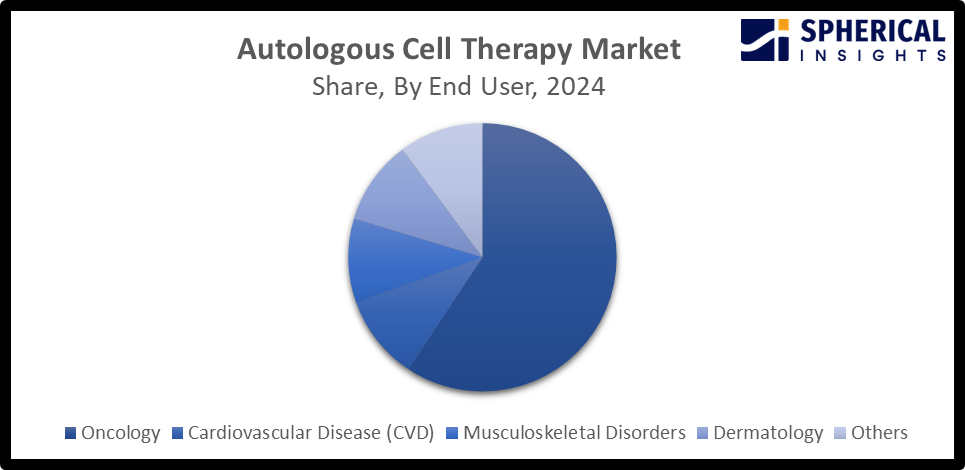
- By therapeutic area, the oncology segment dominates the market with about 85.0% share, while the musculoskeletal segment is anticipated to grow at a significant CAGR during the forecast period.
According to a research report published by Spherical Insights and Consulting, the Global Autologous Cell Therapy Market size was worth around USD 5.39 Billion in 2024 and is predicted to grow to around USD 45.40 Billion by 2035 with a compound annual growth rate (CAGR) of 21.38% from 2025 and 2035. The market for autologous cell therapy has a number of opportunities to grow due to the expanding new therapeutic areas and the development of advanced, localized, point-of-care delivery systems.
Key Trends in Autologous Cell Therapy Market –
- Advancements in CAR-T cell therapies for oncology
- Shift towards automated, closed-loop manufacturing to reduce
- Expansion into autoimmune diseases, decentralized “point-of-care” production
- Increasing use of AI for enhancing manufacturing
Market Overview
The global autologous cell therapy industry encompasses the personalized treatments using a patient’s own cells, which are cultured and expanded outside the body, and reintroduced into the donor to combat diseases like cancer. Autologous cell therapy is garnering attention for its potential to drive significant biopharma interest and investment in the space. Innovation in cell engineering is essentially unlocking the full potential of autologous cell therapy, bending the cost culture to enable developers to bring the massive therapeutic potential of this class to more patients who could realize life-changing benefits. Furthermore, major players’ growing R&D expenditures and industrial expansion are promoting market innovation. For instance, in March 2026, AstraZeneca plans to establish a commercial cell therapy manufacturing and supply base alongside a dedicated innovation center in Shanghai, expanding its end-to-end cell therapy capabilities in China.
Report Coverage
This research report categorizes the autologous cell therapy market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the autologous cell therapy market. Recent market developments and competitive strategies such as expansion, type launch, development, partnership, merger, and acquisition have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the autologous cell therapy market.
Global Autologous Cell Therapy Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 5.39 Billion |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | CAGR of 21.38% |
| 2035 Value Projection: | USD 45.40 billion |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 240 |
| Tables, Charts & Figures: | 110 |
| Segments covered: | By Therapy Type, By Therapeutic Area |
| Companies covered:: | Bluebird bio, Inc., Novartis, Gilead Sciences, Inc. (Kite), Bristol-Myers Squibb Company, Pharmicell Co., Ltd., Johnson & Johnson (Janssen Global Services, LLC.), JW Therapeutics, Holostem S.r.l., Vertex Pharmaceuticals, Inc., Autolus therapeutics, and Others |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
Increasing demand for personalized and precision medicine
While using autologous stem cells, identification of the most appropriate cell sources for different potential applications, development of appropriate clinical-grade methodologies and large-scale, well-controlled clinical trials are essential for assessing the safety and value of cell-based therapies. Driven by personalized CAR-T cell therapies and advanced CRISPR gene editing technologies, the autologous cell therapy market is projected to grow.
Approval of several autologous cell therapies with increasing investment
Autologous cell therapy product launches, namely CARVYKTI (ciltacabtagene autoleucel), Talicabtagene Autoleucel, Sartorius Eveo platform, focused on personalized CAR-T treatments for cancers like B-NHL and multiple myeloma, are accelerating, thereby propelling the autologous cell therapy market. Further, an increasing investment by key industry players like Kite Pharma and Obsidian Therapeutics for improving scalability and reducing vein-to-vein time aids in propelling the market growth.
Restraining Factors
Increased cost and need for specialized training
An increased treatment cost, complex, patient-specific manufacturing processes, and intricate logistics are the factors restraining the autologous cell therapy market. Further, the limited accessibility in lower-income regions and reimbursement issues are challenging the market.
Market Segmentation
The Autologous Cell Therapy Market share is classified into therapy type and therapeutic area.
- The non-stem cell therapies segment dominated the market with about 90.0% share in 2024 and is projected to grow at a substantial CAGR during the forecast period.
Based on the therapy type, the autologous cell therapy market is divided into stem cell therapies, non-stem cell therapies, and others. Among these, the non-stem cell therapies segment dominated the market with about 90.0% share in 2024 and is projected to grow at a substantial CAGR during the forecast period. This is due to increasing regulatory approvals for autologous non-stem cell-based therapies. In October 2023, NexCAR 19 is an autologous CAR T cell therapy developed by an Indian biopharmaceutical startup, ImmunoACT, highlighting ongoing developments in autologous non-stem-based therapies. While the stem cell therapies segment is a significantly growing segment in the autologous cell therapy market, it is driven by its use to treat Hodgkin’s lymphoma, myeloma, non-Hodgkin’s lymphoma, and plasma cell disorders.
- The oncology segment accounted for the largest market share of about 85.0% in 2024 and is projected to grow at a substantial CAGR during the forecast period.
Based on the therapeutic area, the autologous cell therapy market is divided into oncology, cardiovascular disease (CVD), musculoskeletal disorders, dermatology, and others. Among these, the oncology segment accounted for the largest market share of about 85.0% in 2024 and is projected to grow at a substantial CAGR during the forecast period. Immune-oncology cell therapies are one of the fastest advancing technologies with huge R&D activities, contributing to driving the segmental market growth. Further, modified, autologous T-cell immunotherapies are transforming the treatment of hematologic malignancies with several approvals of chimeric antigen receptor T-cell therapies for treating lymphoma, leukaemia, and multiple myeloma. On the other side, the musculoskeletal segment is a growing segment driven by an increasing prevalence of musculoskeletal disorders along with an surging advancements in stem cell technologies.
Get more details on this report -
Regional Segment Analysis of the Autologous Cell Therapy Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the Autologous Cell Therapy market over the predicted timeframe.
North America is anticipated to hold the largest share of about 52.0% in the Autologous Cell Therapy market over the predicted timeframe. The market ecosystem in North America is strong, due to the increasing approvals of cell and gene therapy. For instance, in June 2024, C3i received regulatory approval to produce cell therapies in Canada. The demand for Autologous Cell Therapy has been driven by the region's increasing demand for personalized treatments, increased cancer prevalence, chronic diseases, and investment in regenerative medicine. The U.S. is leading the North America Autologous Cell Therapy market, accounting for 85.0% share, due to the approval of CAR-T cell therapies, the presence of key companies, and increased adoption of CAR-T and personalized therapies.
Get more details on this report -
Asia Pacific is expected to grow at a rapid CAGR of approximately 23.0% in the Autologous Cell Therapy market during the forecast period. The Asia Pacific area has a thriving market for Autologous Cell Therapy due to the growing prevalence of chronic diseases, strong government funding, and fast-track regulatory approvals. Due to their government-backed initiatives, clinical trial activity, and supportive regulatory frameworks, especially in China, Japan, and South Korea, is propelling the market. China accounts for the largest revenue share in the region, with nearly 40% contribution in the region. This is due to strong government support for regenerative medicine and cell therapy, the expanding biotech industry, and increased clinical trial activity.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the autologous cell therapy market, along with a comparative evaluation primarily based on their type of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes type development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Bluebird bio, Inc.
- Novartis
- Gilead Sciences, Inc. (Kite)
- Bristol-Myers Squibb Company
- Pharmicell Co., Ltd.
- Johnson & Johnson (Janssen Global Services, LLC.)
- JW Therapeutics
- Holostem S.r.l.
- Vertex Pharmaceuticals, Inc.
- Autolus therapeutics
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting And Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
In January 2026, Cipla Limited, through its subsidiary Medpro Pharmaceutica, entered into an exclusive license and supply agreement with Immunoadoptive Cell Therapy Private Limited (ImmunoACT).
In November 2025, Aspen Neuroscience Lands $115M to Accelerate Personalised Parkinson’s Cell Therapy. The rise of powers trials of ANPD001, the leading autologous iPSC-derived therapy for moderate to advanced Parkinson’s.
In September 2025, Aspen Neuroscience, Inc., a clinical-stage biotechnology company pioneering autologous regenerative therapies, announced the initiation of Cohort 3 in its ASPIRO Phase 1/2a clinical trial of ANPD001, the company's personalized dopaminergic neuronal precursor cell (DANPC) therapy for moderate to advanced Parkinson's disease (PD).
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the Autologous Cell Therapy Market based on the below-mentioned segments:
Global Autologous Cell Therapy Market, By Therapy Type
- Stem Cell Therapies
- Non-Stem Cell Therapies
- Others
Global Autologous Cell Therapy Market, By Therapeutic Area
- Oncology
- Cardiovascular Disease (CVD)
- Musculoskeletal Disorders
- Dermatology
- Others
Global Autologous Cell Therapy Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
-
1. What is the market size of the autologous cell therapy market?The global autologous cell therapy market size is expected to grow from USD 5.39 Billion in 2024 to USD 45.40 Billion by 2035, at a CAGR of 21.38% during the forecast period 2025-2035.
-
2. Which region holds the largest share of the autologous cell therapy market?North America is anticipated to hold the largest share of the autologous cell therapy market over the predicted timeframe.
-
3. What is the forecasted CAGR of the Global Autologous Cell Therapy Market from 2024 to 2035?The market is expected to grow at a CAGR of around 21.38% during the period 2024–2035.
-
4. Who are the top companies that are involved in the Global Autologous Cell Therapy Market?Key players include Bluebird bio, Inc., Novartis, Gilead Sciences, Inc. (Kite), Bristol-Myers Squibb Company, Pharmicell Co., Ltd., Johnson & Johnson (Janssen Global Services, LLC.), JW Therapeutics, Holostem S.r.l., Vertex Pharmaceuticals, Inc., and Autolus Therapeutics.
-
5. What are the main drivers in the autologous cell therapy market?An increasing demand for personalized and precision medicine as well as approval of several autologous cell therapies with increasing investment, are major market growth drivers of the Autologous Cell Therapy market.
-
6. What challenges are limiting the adoption of Autologous Cell Therapy?Factors like the increased cost and need for specialized training, as well as limited accessibility and reimbursement issues, remain key restraints in the autologous cell therapy market.
-
7. What are the key trends in the autologous cell therapy market?The surging advancements in CAR-T cell therapies for oncology, a shift towards automated, closed-loop manufacturing to reduce costs, expansion into autoimmune diseases, decentralized “point-of-care” production, and an increasing use of AI for enhancing manufacturing are major key trends in the autologous cell therapy market.
Need help to buy this report?