Global Viral Vector and Plasmid DNA Testing Services Market To Surpass USD 602 Million By 2030 | CAGR 25.5 %
Category: HealthcareGlobal Viral Vector and Plasmid DNA Testing Services Market worth $ 602 million by 2030
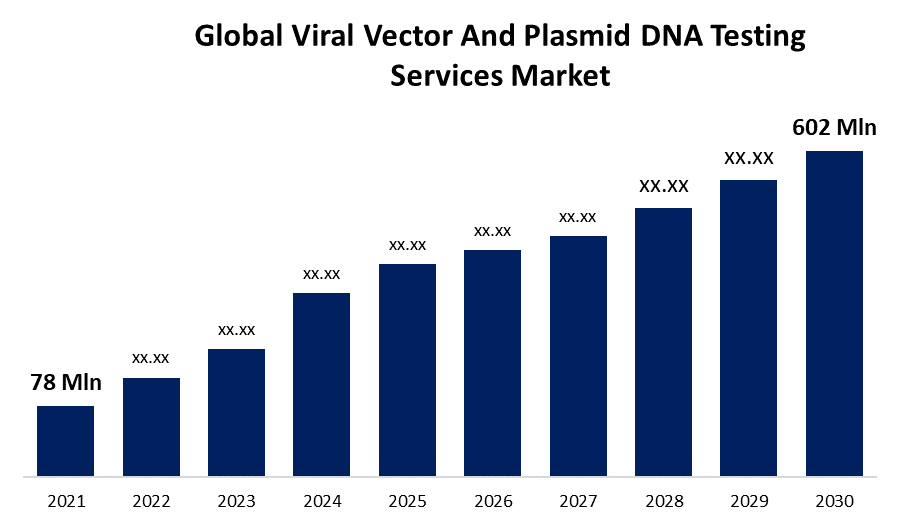
According to a research report published by Spherical Insights & Consulting, the Global Viral Vector and Plasmid DNA Testing Services Market Size to grow from USD 78 million in 2021 to USD 602 million by 2030, at a Compound Annual Growth Rate (CAGR) of 25.5% during the forecast period.
Get more details on this report -
The Viral vector and plasmid DNA testing services market has grown due to increased research and development. In addition, the wide use of these tools in delivering genetic materials into cells also expands the market growth.
Browse 48 market data Tables and 58 Figures spread through 210 Pages and in-depth TOC on “Global Viral Vector And Plasmid DNA Testing Services Market Size, Share, and COVID-19 Impact Analysis, By Testing Services (Safety, Genetic Characterization, Purity, Identity, and Potency), By End-User (Research Organizations and Pharmaceutical and Biotechnological Companies), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2021 – 2030.”
View a detailed Table of Content here– https://www.sphericalinsights.com/reports/viral-vector-and-plasmid-dna-testing-services-market
The COVID-19 pandemic has made an adverse impact on credit portfolios. There has been an unprecedented rise in unemployment and disruption in economic activity, putting a strain on the solvency of customers and companies. Central banks have taken a proactive approach by injecting liquidity into the market by lowering interest rates and asset purchase programs. Managing and monitoring credit, market, liquidity, and operational risk across financial markets were hard enough with ongoing geopolitical tensions, international trade wars, and the occasional hurricanes and earthquakes. The current pandemic situation has forced chief risk officers and their teams to recalibrate old assumptions and models used to manage and monitor risk. COVID-19’s global impact has shown that interconnectedness plays an important role in international cooperation. As a result, many governments started rushing toward identifying, evaluating, and procuring reliable solutions powered by AI.
The Safety segment to account for the largest market size during the forecast period
Based on the testing services, the global Viral vector and plasmid DNA testing services market is categorized into Safety, Genetic Characterization, Purity, Identity, and Potency. The Safety segment to account for the largest market size during the forecast period. Because manufacturers are launching more goods and services, the market is likely to develop as a result. To increase the availability of its clinical testing services in the Asia Pacific market, Merck KGaA, for instance, opened the GMP BioReliance biosafety testing laboratory in Singapore in September 2018. Additionally, LabCorp's Covance drug development division introduced a portfolio of cell and gene therapy development products in January 2020. The remedy is created to hasten the advancement of gene and cell treatment. Under this, Labcorp provides clinical and preclinical solutions, including safety evaluation and bioanalysis testing and services.
The research organizations segment to hold a higher CAGR during the forecast period
Based on end-user, the Viral vector and plasmid DNA testing services market is categorized into Research Organizations and Pharmaceutical and Biotechnological Companies. The research organizations segment to hold a higher CAGR during the forecast period. Due to the high demand for viral vectors for conducting research, it is anticipated that the growing involvement of scientific communities in gene and cell therapy research would boost the demand for viral vectors. In addition, the market for viral vectors and plasmid DNA testing is seeing the emergence of research institutions as major end-users, which is helping to drive the development of sophisticated drugs and an increasing number of gene therapy-based R&D initiatives. One of these companies, Abeona Therapeutics, for instance, is exploring AAV9-based gene therapies for CLN1 and CLN3 illnesses. It will therefore encourage market expansion.
Asia-Pacific to hold a higher CAGR during the forecast period.
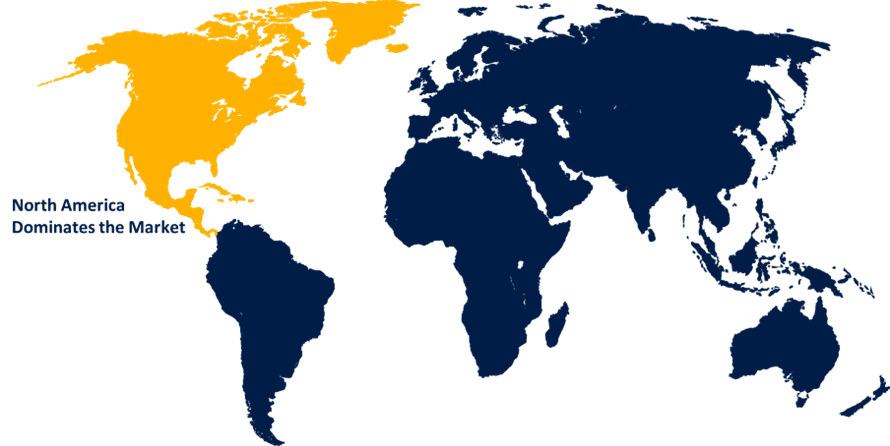
The Global Viral Vector and Plasmid DNA Testing Services Market has been segmented into five major regions: North America, Europe, Asia-Pacific, South America, The Middle East and Africa. North America is estimated to account for the highest market share in 2022. North America is expected to be the largest market. The existence of a sizable number of centers and institutes involved in the R&D of advanced medicines is one of the key factors that has contributed to the huge share of this regional market. The federal agencies' investments in the region's cell therapy research base are expected to boost the market's expansion in North America.
Get more details on this report -
Major vendors in the Global Viral Vector and Plasmid DNA Testing Services Market include
Charles River Laboratories, Inc., WuXi AppTec Co., Ltd., Cobra Biologics and Pharmaceutical Services, Merck KgaA, Lonza, Eurofins Scientific, FinVector Vision Therapies, Advanced Bioscience Laboratories, Inc., Takara Bio Inc., ViruSure GmbH, and Genezen Laboratories.
Contact Us: https://www.sphericalinsights.com/contact-us
Frequently Asked Questions (FAQ)
-
What is the market size of the Viral Vector and Plasmid DNA Testing Services market?As per Spherical Insights, the size of the Viral Vector and Plasmid DNA Testing Services market was valued at USD 78 million in 2022 to USD 602 million by 2030.
-
What is the market growth rate of the Viral Vector and Plasmid DNA Testing Services market?The Viral Vector and Plasmid DNA Testing Services market is growing at a CAGR of 25.5% from 2022 to 2030.
-
Which country dominates the Viral Vector and Plasmid DNA Testing Services market?North America emerged as the largest market for Viral Vector and Plasmid DNA Testing Services.
-
Who are the key players in the Viral Vector and Plasmid DNA Testing Services market?Key players in the Viral Vector and Plasmid DNA Testing Services market are Charles River Laboratories, Inc., WuXi AppTec Co., Ltd., Cobra Biologics and Pharmaceutical Services, Merck KgaA, Lonza, Eurofins Scientific, FinVector Vision Therapies, Advanced Bioscience Laboratories, Inc., Takara Bio Inc., ViruSure GmbH, and Genezen Laboratories.
-
Which factor drives the growth of the Viral Vector and Plasmid DNA Testing Services market?Rise in R&D is expected to drives the market's growth over the forecast period.
Need help to buy this report?