Global Lipid Nanoparticle CDMO Market Size is Projected to Reach USD 1405.5 Million by 2035 | CAGR of 14.22%
Category: HealthcareGlobal Lipid Nanoparticle CDMO Market Size is projected to reach USD 1405.5 Million by 2035
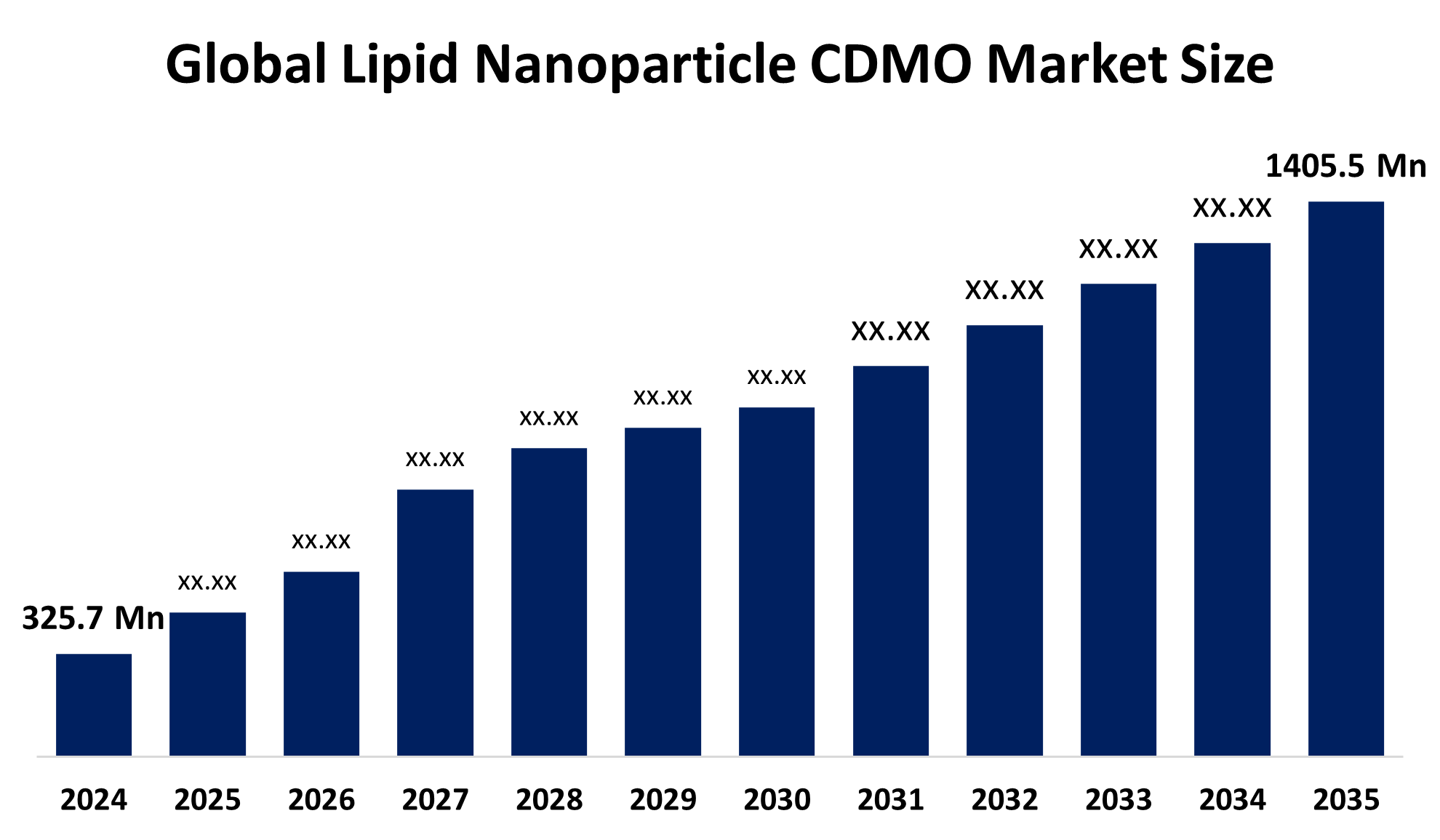
According to a research report published by Spherical Insights & Consulting, the Global Lipid Nanoparticle CDMO Market is expected to grow from USD 325.7 million in 2024 to USD 1405.5 million by 2035, at a CAGR of 14.22% during the forecast period 2025-2035.
Get more details on this report -
Browse 210 Market Data Tables And 45 Figures Spread Through 190 Pages and In-Depth TOC On the "Global Lipid Nanoparticle CDMO Market Size, Share, and COVID-19 Impact Analysis, By Service Type (Development & Process Development, LNP Formulation & Encapsulation, Analytical & Quality Control, cGMP Drug Product Manufacturing, and Fill-Finish Lyophilization), By Application (Infectious Disease Vaccines, Oncology Therapeutics, Rare/Genetic & Metabolic Disorders, and Other Therapeutics), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 – 2035." Detailed Report Decription Here : https://www.sphericalinsights.com/reports/lipid-nanoparticle-cdmo-market
The lipid nanoparticle CDMO market refers to the worldwide industry that involves specialized service providers developing and manufacturing LNPs for pharmaceutical companies, which is driven by the upsurging need for advanced delivery systems in mRNA vaccines, oncology, and gene therapy. Biopharma industries are outsourcing formulation and manufacturing of lipid nanoparticle based medications, providing specialized services for designing, formulating, analyzing, and manufacturing lipid-based delivery vehicles for mRNA, siRNA, and other nucleic acid therapeutics. It is anticipated that continued developments in nanotechnology and the growing uses of LNPs in cancer and gene therapy quicken market growth. Support for government-backed investments and strategic partnerships is surging for mRNA vaccines and therapeutic production, which contributes to propelling market growth. On the contrary, increased, complex manufacturing costs, lack of specialized technical expertise, and regulatory complexities are restraining the lipid nanoparticle CDMO market.
The development & process development segment dominated the market with the largest share of about 35.2% in 2024 and is projected to grow at a substantial CAGR during the forecast period.
Based on the service type, the lipid nanoparticle CDMO market is divided into development & process development, LNP formulation & encapsulation, analytical & quality control, cGMP drug product manufacturing, and fill-finish lyophilization. Among these, the development & process development segment dominated the market with the largest share of about 35.2% in 2024 and is projected to grow at a substantial CAGR during the forecast period. The segment includes end-to-end development, process optimization, and cGMP manufacturing for advanced therapeutics. cGMP drug-product manufacturing remains the largest pool of approved vaccines and therapeutics.
The oncology therapeutics segment accounted for the largest share of about 40.5% in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period.
Based on the application, the lipid nanoparticle CDMO market is divided into infectious disease vaccines, oncology therapeutics, rare/genetic & metabolic disorders, and other therapeutics. Among these, the oncology therapeutics segment accounted for the largest share of about 40.5% in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. CDMOs provide important expertise in formulation development, process scaling, and GMP-compliant manufacturing for complex, 10-1000nm, lipid-based systems for specific tumor sites.
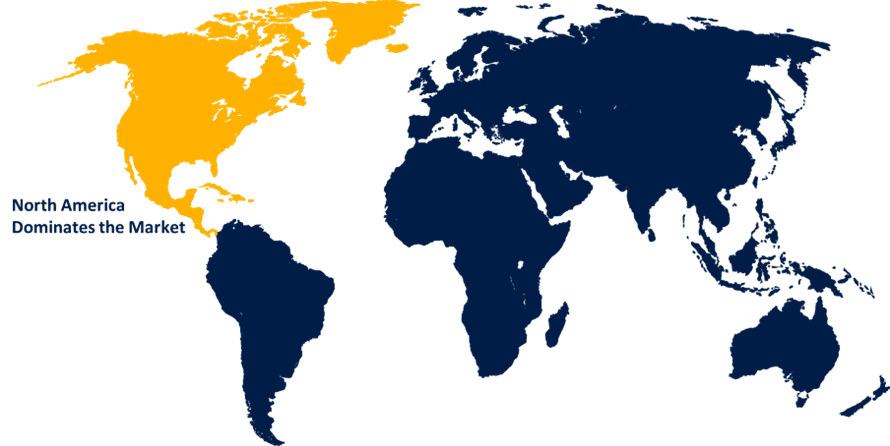
North America is expected to hold the majority share of the global lipid nanoparticle CDMO market during the forecast period.
Get more details on this report -
North America is expected to hold the majority share of around 45.5% in the global lipid nanoparticle CDMO market during the forecast period. The U.S. accounts for the largest portion of about 43.0% share within the North America region for the lipid nanoparticle CDMO market, driven by the presence of a strong biopharmaceutical ecosystem, investments in mRNA/LNP technologies, and a supportive regulatory environment and funding programs. North America has a robust market ecosystem that includes both innovative research & development activities. Further, the region's increasing demand for mRNA-based vaccines and therapeutics, along with an increased prevalence of chronic/infectious diseases requiring targeted delivery and R&D investment, is contributing to driving the market.
Asia Pacific is anticipated to grow at the fastest pace of around 15.3% CAGR in the global lipid nanoparticle CDMO market during the forecast period. China is leading the Asia Pacific lipid nanoparticle CDMO market, with about 15.0% CAGR value, owing to the increasing investment in mRNA and nucleic acid therapeutics. In the Asia Pacific lipid nanoparticle CDMO market, there is a rise in healthcare investments and expanding biopharmaceutical manufacturing capabilities, and advanced research facilities in countries like Japan and South Korea. Governments' active support of the regulatory frameworks, and long-term funding, along with the presence of specialized and large-scale contract manufacturers are supporting the market
Major vendors in the global lipid nanoparticle CDMO market are AGC Biologics, CordenPharma, Curia, Eurogentec, Evonik Health Care, Fujifilm Toyama Chemical, Lonza, PCI Pharma Services, Polymun Scientific, Porton Advanced, Recipharm, Rentschler Biopharma, Samsung Biologics, Thermo Fisher Scientific, and Others.
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Key Market Development
- In February 2026, Contract development and manufacturing organization (CDMO) Cellipont Bioservices announced a partnership with Soter Bio, a CDMO specializing in the development of lipid nanoparticles for RNA therapies. The companies would collaborate on integrated manufacturing techniques to produce complex cell therapies.
- In September 2025, Evonik, a global specialty chemicals company, and Ethris, a clinical-stage biotechnology company pioneering next-generation RNA therapeutics and vaccines, entered a strategic collaboration to develop and market a novel lipid nanoparticle (LNP) platform for nucleic acid delivery.
- In March 2025, Amaran Biotechnology Inc., a subsidiary of OBI Pharma specialising in Contract Development and Manufacturing Organization (CDMO) services for next-generation pharmaceuticals, signed a Memorandum of Understanding (MoU) with Nippon Fine Chemical Co., Ltd., and its Taiwan subsidiary, Zillion Fine Chemicals International Co., Ltd.
- In July 2024, Agilent is upping CDMO presence through $925m Biovectra buy. The deal announced today sees Agilent Technologies bolster its current oligonucleotide and CRISPR-based therapeutics manufacturing services with the addition of contract development and manufacturing organization (CDMO) Biovectra.
- In June 2024, CordenPharma partnered with Certest to develop Ionizable Lipids for LNP formulations. Switzerland-based CordenPharma, a contract development and manufacturing organization, announced that it has partnered with Certest, a Spain-based company focused on lipid nanoparticle (LNP)-based drug delivery and API synthesis, to develop a portfolio of ionizable lipids for LNP formulations.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the lipid nanoparticle CDMO market based on the below-mentioned segments:
Global Lipid Nanoparticle CDMO Market, By Service Type
- Development & Process Development
- LNP Formulation & Encapsulation
- Analytical & Quality Control
- cGMP Drug Product Manufacturing
- Fill-Finish Lyophilization
Global Lipid Nanoparticle CDMO Market, By Application
- Infectious Disease Vaccines
- Oncology Therapeutics
- Rare/Genetic & Metabolic Disorders
- Other Therapeutics
Global Lipid Nanoparticle CDMO Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
About the Spherical Insights & Consulting
Spherical Insights & Consulting is a market research and consulting firm which provides actionable market research study, quantitative forecasting and trends analysis provides forward-looking insight especially designed for decision makers and aids ROI.
Which is catering to different industry such as financial sectors, industrial sectors, government organizations, universities, non-profits and corporations. The company's mission is to work with businesses to achieve business objectives and maintain strategic improvements.
CONTACT US:
For More Information on Your Target Market, Please Contact Us Below:
Phone: +1 303 800 4326 (the U.S.)
Phone: +91 90289 24100 (APAC)
Email: inquiry@sphericalinsights.com, sales@sphericalinsights.com
Contact Us: https://www.sphericalinsights.com/contact-us
Need help to buy this report?