
Top 20 Companies in Global Medical Device Labeling Market (2025–2035): Expert View by Spherical Insights
RELEASE DATE: Apr 2026 Author: Spherical InsightsRequest Free Sample Speak to Analyst
Introduction
The global Medical Device Labeling market refers to the systems and solutions used to provide essential product information, identification, and regulatory compliance details on medical devices. Strict regulations which include the Unique Device Identification (UDI) system and the rising production of medical devices and the increasing need for patient safety and traceability drive market expansion. The market is further supported by growing adoption of digital and smart labeling technologies which include RFID and barcode systems. The emerging markets along with advanced labeling solutions which enhance supply chain visibility present new business opportunities. Governments worldwide are strengthening labeling standards; for instance, the U.S. Food and Drug Administration mandates UDI compliance, enhancing transparency and reducing errors across healthcare systems.
In December 2025, Thailand’s Ministry of Public Health officially launched a new medical device labeling regulation, which will be fully implemented in June 2026. The update introduces structured labeling requirements, electronic instructions for use (e-IFU), and clearer classification systems, improving compliance and digital adoption in labeling practices.
Navigate Future Markets with Confidence: Insights from Spherical Insights LLP
The insights presented in this blog are derived from comprehensive market research conducted by Spherical Insights LLP, a trusted advisory partner to leading global enterprises. Backed by in-depth data analysis, expert forecasting, and industry-specific intelligence, our reports empower decision-makers to identify strategic growth opportunities in fast-evolving sectors. Clients seeking detailed market segmentation, competitive landscapes, regional outlooks, and future investment trends will find immense value in the full report. By leveraging our research, businesses can make informed decisions, gain a competitive edge, and stay ahead in the transition toward sustainable and profitable solutions.
Unlock exclusive market insights—Download the Brochure now and dive deeper into the future of the Medical Device Labeling Market.
Market Segmentation
Global Medical Device Labeling Market Size, Share, By Label Type (Glue-Applied Labels, Pressure-Sensitive Labels, In-Mold Labels, Sleeve Labels, and Others), By Material Type (Plastic, Paper, and Others), and By Application (Monitoring & Diagnostic Equipment, Disposable Consumables, and Therapeutic Equipment), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 – 2035
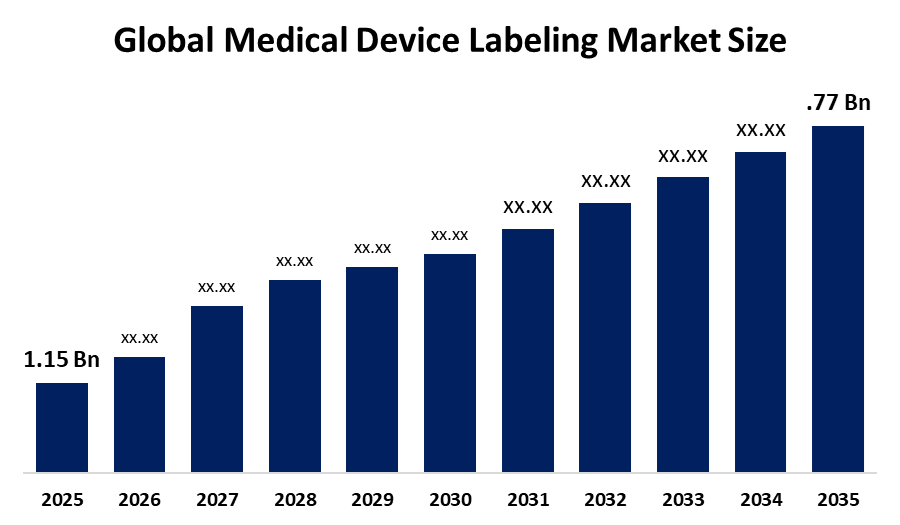
Medical Device Labeling Market Size & Statistics
- The Market Size for Medical Device Labeling Was Estimated to be worth USD 1.15 Billion in 2025.
- The Market is Going to Expand at a CAGR of 4.41% between 2025 and 2035.
- The Global Medical Device Labeling Market Size is anticipated to reach USD 1.77 Billion by 2035.
- North America is expected to generate the highest demand during the forecast period in the Medical Device Labeling Market
- Asia Pacific is expected to grow the fastest during the forecast period in the Medical Device Labeling Market.
Regional growth and demand
Asia Pacific is expected to grow the fastest during the forecast period in the Medical Device Labeling market. Asia Pacific is expected to grow fastest due to expanding healthcare infrastructure, rising medical device manufacturing, and increasing regulatory standardization. Countries like China, India, and Japan are strengthening labeling regulations and adopting global standards such as UDI systems. Rapid growth in healthcare investments, medical exports, and digital transformation is boosting demand for advanced labeling solutions. Furthermore, increasing awareness of patient safety and the adoption of cost-effective labeling technologies are supporting market expansion, making the region a key growth hub for medical device labeling in the coming years.
North America is expected to generate the highest demand during the forecast period in the Medical Device Labeling market. North America is expected to generate the highest demand due strict regulatory frameworks and advanced healthcare infrastructure. Authorities such as the U.S. Food and Drug Administration mandate systems like Unique Device Identification (UDI), increasing the need for accurate and compliant labeling. The region also has a high volume of medical device manufacturing and strong adoption of advanced technologies such as RFID and barcode labeling. Additionally, growing emphasis on patient safety, traceability, and efficient recall systems further drives demand for sophisticated labeling solutions across the healthcare sector.
Unlock exclusive market insights—Download the Brochure now and dive deeper into the future of the Medical Device Labeling Market.
Ready to lead the Medical Device Labeling Market ?
Discover the regional trends and growth factors shaping the industry. We’re here to assist with expert, personalized data.
Call +1 303 800 4326 or Send us a message for a personalized consultation.
Top 10 Trends in the Medical Device Labeling Market
- Adoption of digital and electronic labeling systems
- Expansion of Unique Device Identification (UDI) requirements
- Integration of smart technologies (QR, RFID, NFC)
- Increasing focus on regulatory compliance and standardization
- Growth of multilingual and global labeling solutions
- Use of automated label management systems
- Shift toward sustainable and eco-friendly labeling materials
- Implementation of track-and-trace capabilities
- Advancements in high-quality printing technologies
- Emphasis on patient-friendly and clear label design
- Adoption of digital and electronic labeling systems
The medical device labeling market experiences transformations through the implementation of digital and electronic labeling systems which replace traditional paper-based instructions through the use of QR codes and mobile applications and web-based platforms. The system provides organizations with the ability to access real-time information and display content in multiple languages while enhancing patient safety measures. The EU MDR regulatory framework increasingly allows electronic labeling through its requirements which specify digital compliance for particular devices. The European Commission supports digital data systems that connect to device identification databases because these systems enhance system visibility and user accessibility.
- Expansion of Unique Device Identification (UDI) Requirements
The medical device labeling industry experiences its primary regulatory development through the expansion of UDI requirements which lead to market growth. UDI systems require every device to carry a unique code on labels and packaging which improves traceability and patient safety. The FDA has established UDI compliance through its recent updates of guidance materials together with its inspection programs. In 2026, FDA expanded compliance programs to emphasize labeling and UDI tracking across the entire product lifecycle. This ensures better recall management and reduces medical errors, making UDI a global standard adopted across regions including the US and Europe.
- Integration of smart technologies (QR, RFID, NFC)
Smart labeling technologies which include QR codes and RFID and NFC, have become common, medical device labels to enhance traceability and user interaction. The technologies provide users with immediate access to product details and usage information and authentication data through digital channels. The present regulatory frameworks permit automatic identification and data capture (AIDC) technologies to function as components of UDI systems. The U.S. Food and Drug Administration consider machine-readable identifiers necessary for both electronic health records and supply chain systems. The system functions through an integrated solution that boosts operational productivity while decreasing counterfeit threats and enabling sophisticated healthcare data administration.
- Increasing focus on regulatory compliance and standardization
Regulatory compliance functions as the primary force that drives development in the medical device labeling market because global authorities enforce strict labeling requirements. The U.S. Food and Drug Administration and international regulations such as EU MDR require detailed labeling, including device identification, safety information, and standardized formats. The FDA issued more than 600 warning letters during 2025, which included multiple letters that addressed regulatory and labeling compliance violations, because the agency viewed compliance as critical. The regulations establish requirements for patient safety and product transparency and quality consistency, which drive manufacturers to implement standardized labeling systems throughout the world.
- Growth of multilingual and global labeling solutions
The demand for multilingual labeling is increasing due to the global distribution of medical devices and diverse regulatory requirements. Manufacturers must provide labels in multiple languages to comply with regional laws and ensure patient safety. Under the EU MDR framework supported by the European Commission, labeling must include clear translations for all member states. This requirement drives the adoption of advanced labeling software and automated translation systems
Empower your strategic planning:
Stay informed with the latest industry insights and market trends to identify new opportunities and drive growth in the medical device labeling market. To explore more in-depth trends, insights, and forecasts, please refer to our detailed report.
Top 20 Companies Leading the Medical Device Labeling Market
- Avery Dennison Corporation
- 3M Company
- CCL Industries Inc.
- Zebra Technologies Corporation
- Loftware Inc.
- Seagull Scientific Inc.
- SATO Holdings Corporation
- Honeywell International Inc.
- Lintec Corporation
- UPM Raflatac
- Schreiner Group
- TEKLYNX International
- NiceLabel
- WS Packaging Group Inc.
- FLEXcon Company Inc.
- Mactac LLC
- Systech International
- Impinj Inc.
- Computype Inc.
- Amcor plc
Unlock exclusive market insights—Download the Brochure now and dive deeper into the future of the Medical Device Labeling Market.
- Avery Dennison Corporation
Headquarters: Mentor, Ohio, USA
Avery Dennison Corporation operates as a worldwide authority in the development of labeling solutions and packaging materials through its creation of pressure-sensitive labels and RFID technologies and digital identification systems. The company has established operations in 50 different countries which employ about 35000 workers to provide services for various sectors including healthcare and retail and logistics and automotive industries. The organization works to establish links between actual products and their corresponding digital information which leads to better supply chain operations and product visibility. The product allows businesses to monitor their items better while building their brand identity and fostering customer relationships. Avery Dennison functions as an essential component of the medical device labeling industry by providing advanced solutions which enable hospitals to meet their regulatory obligations while safeguarding patient health.
- 3M Company
Headquarters: Saint Paul, Minnesota, USA
3M Company operates as an international technology and manufacturing organization which provides solutions across multiple sectors including healthcare and safety and electronics and industrial markets. The company creates various products which include medical tapes and labeling materials and identification solutions that healthcare facilities use. The healthcare division of the organization develops innovative materials and technologies to enhance patient care results. 3M provides medical device manufacturers with high-quality labeling solutions which meet industry standards through its operations that extend across different areas.
- CCL Industries Inc.
Headquarters: Toronto, Canada
CCL Industries Inc. which operates as one of the world's largest label manufacturers, provides various labeling and packaging products to different industries including the healthcare and pharmaceutical sectors. The company operates numerous manufacturing facilities globally and provides advanced labeling technologies such as RFID, shrink sleeves, and specialty labels. The products enable businesses to identify their products, build their brand, and meet their legal requirements. Multinational companies can be served by CCL which operates worldwide and offers various products from its extensive range. The company plays a significant role in the medical device labeling market by delivering high-quality, compliant labeling solutions for critical healthcare applications.
- Zebra Technologies Corporation
Headquarters: Lincolnshire, Illinois, USA
Zebra Technologies Corporation develops automatic identification and data capture technologies which include barcode printers and RFID systems and mobile computing solutions. The company provides tools that enable real-time visibility and tracking of assets across healthcare and supply chain environments. The hospital and medical device manufacturing industries use its solutions for labeling, tracking, and data management purposes. Zebra’s technologies improve medical device labeling through their capacity to boost performance and minimize mistakes while providing better product traceability. The company helps healthcare providers achieve patient safety and regulatory compliance through its worldwide operations and digital expertise.
- Loftware Inc.
Headquarters: Portsmouth, New Hampshire, USA
Loftware Inc. provides enterprise labeling and artwork management software solutions to businesses. The company helps organizations design and manage their labeling systems which must meet international standards like UDI and healthcare regulations. The company offers cloud-based labeling systems that enable businesses to create and manage their labels throughout their worldwide operations. Medical device manufacturers use Loftware solutions to improve their labeling procedures while decreasing mistakes and achieving regulatory requirements. The company leads the changing medical device labeling industry through its dedication to digital transformation and automation.
Are you ready to discover more about the medical device labeling market?
The report provides an in-depth analysis of the leading companies operating in the global medical device labeling market. It includes a comparative assessment based on their product portfolios, business overviews, geographical footprint, strategic initiatives, market segment share, and SWOT analysis. Each company is profiled using a standardized format that includes:
Company Profiles
- Avery Dennison Corporation
- Business Overview
- Company Snapshot
- Products Overview
- Company Market Share Analysis
- Company Coverage Portfolio
- Financial Analysis
- Recent Developments
- Merger and Acquisitions
- SWOT Analysis
- 3M Company
- CCL Industries Inc.
- Zebra Technologies Corporation
- Loftware Inc.
- Seagull Scientific Inc.
- SATO Holdings Corporation
- Honeywell International Inc.
- Lintec Corporation
- Others.
Conclusion
The global market for medical device labeling sees consistent growth because of new regulatory requirements and increased production of medical devices and the need to improve patient safety and tracking mechanisms. Labeling has become a critical component in ensuring compliance, improving product identification, and reducing medical errors across healthcare systems. The market is undergoing a transformation through the implementation of digital labeling, UDI systems, and smart tracking technologies. The market expansion receives additional support from the internationalization of healthcare and the requirement for products to include multilingual labeling. The market will continue to evolve because of ongoing regulatory changes and technological progress which create new business opportunities for companies that pursue compliance and operational efficiency and digital transformation.
Our Top Reports
Construction Punch List Software Market Size, Growth 2035
China Foodservice Market Size and Growth
Europe LPG Market Size, Shares, Trends to 2035
United Kingdom Swine Vaccines Market Forecasts to 2035
Global Vitamin C Market Size, Growth, Forecast 2035
Unlock exclusive market insights—Download the Brochure now and dive deeper into the future of the Medical Device Labeling Market.
About the Spherical Insights & Consulting
Spherical Insights & Consulting is a market research and consulting firm which provides actionable market research study, quantitative forecasting and trends analysis provides forward-looking insight especially designed for decision makers and aids ROI.
Which is catering to different industry such as financial sectors, industrial sectors, government organizations, universities, non-profits and corporations. The company's mission is to work with businesses to achieve business objectives and maintain strategic improvements.
CONTACT US:
For More Information on Your Target Market, Please Contact Us Below:
Phone: +1 303 800 4326 (the U.S.)
Phone: +91 90289 24100 (APAC)
Email: inquiry@sphericalinsights.com, sales@sphericalinsights.com
Contact Us: https://www.sphericalinsights.com/contact-us
Follow Us: LinkedIn | Facebook | Twitter
Need help to buy this report?