United States Trauma Fixation Devices Market Size, Share, and COVID-19 Impact Analysis, By Product (External Fixators, Internal Fixator), By Material (Stainless Steel, Titanium), By Surgical Site (Upper Extremities, Lower extremities), and United States Trauma Fixation Devices Market Insights Forecasts to 2033
Industry: HealthcareUnited States Trauma Fixation Devices Market Insights Forecasts to 2033
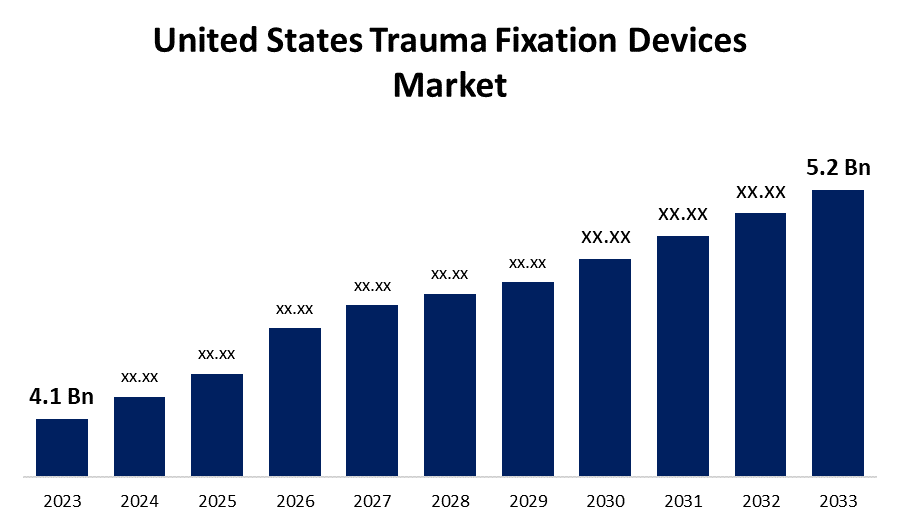
- The United States Trauma Fixation Devices Market Size was valued at USD 4.1 Billion in 2023.
- The Market Size is Growing at a CAGR of 2.4% from 2023 to 2033.
- The United States Trauma Fixation Devices Market Size is Expected to Reach USD 5.2 Billion by 2033.
Get more details on this report -
The United States Trauma Fixation Devices Market Size is expected to reach USD 5.2 Billion by 2033, at a CAGR of 2.4% during the forecast period 2023 to 2033.
Market Overview
Trauma is a physical injury caused by an outside force, such as an accident, dislocation, burns, fractures, strains, or sprain. Trauma fixation is the primary consideration for bone fractures. Trauma fixation area units are used to treat physical traumas. It's an important treatment for bone fractures and tissue degeneration. Trauma fixation devices provide immobilization at the fracture site, reduce the gap, and allow the first bone healing method, callus formation.
Report Coverage
This research report categorizes the market for United States trauma fixation devices market based on various segments and regions and forecasts revenue growth and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the United States trauma fixation devices market. Recent market developments and competitive strategies such as expansion, product launch, and development, partnership, merger, and acquisition have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segments of the United States Trauma Fixation Devices market.
United States Trauma Fixation Devices Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2023 |
| Market Size in 2023: | USD 4.1 Billion |
| Forecast Period: | 2023-2033 |
| Forecast Period CAGR 2023-2033 : | 2.4% |
| 2033 Value Projection: | USD 5.2 Billion |
| Historical Data for: | 2019-2022 |
| No. of Pages: | 200 |
| Tables, Charts & Figures: | 110 |
| Segments covered: | By Product, By Material, By Surgical Site and COVID-19 Impact Analysis. |
| Companies covered:: | Acumed LLC, Bioretec LTD, Depuy Synthes (Johnson & Johnson), Smith & Nephew, Integra LifeSciences, B Braun Melsungen AG, CONMED Corporation, Wright Medical Group N V, Stryker Corporation, Zimmer Biomet Holdings Inc. and Other Key Vendors. |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
Increased morbidity, an aging population, and an increase in road accident and injury cases, as well as high health care costs, pro-government reimbursement policies, and age, are significant factors driving the growth of the trauma fixation market during the forecast period. Furthermore, as related diseases and disorders increase, so do consumer investments and spending in healthcare, accelerating the market's overall growth. Technologically advanced products such as drug-eluting implants, bioresorbable implants, and nano-coated devices are expected to drive market growth. Demand for this specialty has increased significantly as a result of technological advancements, which are expected to create profitable opportunities for the market. Furthermore, the simultaneous conversion of these metal-based products or preparations can result in allergies and infections such as eczema, urticaria, and vasculitis. Metal sensitivity, which can lead to osteoporosis, pain, and aseptic handling of implanted metal hardware, are also expected to limit market growth during the forecast period.
Restraining Factors
Factors such as a lack of trained professionals and technical expertise, as well as issues with these devices, hamper market growth. Furthermore, a lack of awareness, multiple import tariffs on health care products, and high costs may impede market growth during the forecast period.
Market Segment
- In 2023, the internal fixator Fixation Devices segment accounted for the largest revenue share over the forecast period.
Based on the product, the United States trauma fixation devices market is segmented into external fixators, and internal fixator. Among these, the internal fixator segment has the largest revenue share over the forecast period. Internal fixators have an advantage over external fixators in terms of durability, healing time, and bone support. These can be made of a variety of materials, including stainless steel and titanium. Furthermore, advanced materials such as biopolymers provide a turnkey platform for manufacturers.
- In 2023, the titanium segment accounted for the largest revenue share over the forecast period.
Based on the material, the United States trauma fixation devices market is segmented into stainless steel and titanium. Among these, the titanium segment has the largest revenue share over the forecast period. Titanium is widely used by manufacturers to create trauma fixation devices due to its high biocompatibility and low post-operative complications. It also has a lower modulus of elasticity, which reduces the product's rigidity and limits the amount of stress applied to bony structures. Titanium alloys are increasingly being used to make orthopedic and trauma implants because they have a high tensile strength-to-density ratio, osseointegration, corrosion resistance, and are nontoxic.
- In 2023, the lower extremities segment accounted for the largest revenue share over the forecast period.
Based on the surgical site, the United States trauma fixation devices market is segmented into upper extremities and lower extremities. Among these, the lower extremities segment has the largest revenue share over the forecast period. The lower extremities consist of the foot and ankle, thigh, hip and pelvis, lower leg, and knee. The increase is attributed to an increase in the number of people suffering from bone-related injuries, traumatic accidents, and osteoporosis.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the United States trauma fixation devices market along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Acumed LLC
- Bioretec LTD
- Depuy Synthes (Johnson & Johnson)
- Smith & Nephew
- Integra LifeSciences
- B Braun Melsungen AG
- CONMED Corporation
- Wright Medical Group N V
- Stryker Corporation
- Zimmer Biomet Holdings Inc.
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In February 2021, The ExtremiLOCK Lateral Ankle Fusion Plates have been approved by the US FDA. The product is intended to be used to repair osteotomies, fractures, and non-unions of the fibula and tibia. The development of ankle fusion plates has the potential to significantly reduce the amount of time required to treat these injuries.
Market Segment
This study forecasts country revenue from 2022 to 2033. Spherical Insights has segmented the United States trauma fixation devices market based on the below-mentioned segments:
United States Trauma Fixation Devices Market, By Product
- External Fixators
- Internal Fixator
United States Trauma Fixation Devices Market, By Material
- Stainless Steel
- Titanium
United States Trauma Fixation Devices Market, By Surgical Site
- Upper Extremities
- Lower extremities
Need help to buy this report?