Global Pediatric Non-invasive Wearables Market Size, Share, and COVID-19 Impact Analysis, By Monitoring Type (Vitals and Temperature Wearables, SpO2 and Respiratory Wearables, Activity and Sleep Wearables, and Other), By Patient Age Group (Neonates (0-28 Days), Infants (1-12 Months), Children (1-12 Years), and Others), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 - 2035.
Industry: HealthcareGlobal Pediatric Non-invasive Wearables Market Insights Forecasts to 2035
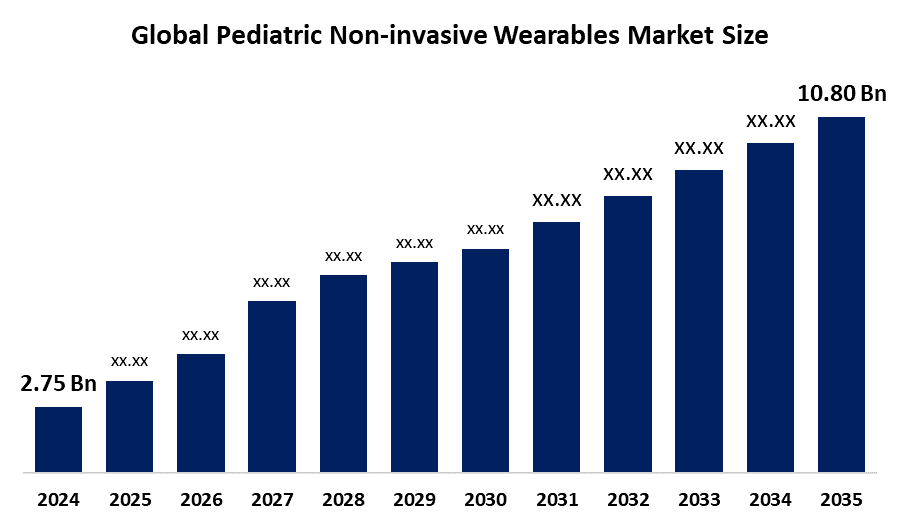
- The Global Pediatric Non-invasive Wearables Market Size Was Estimated at USD 2.75 Billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 13.24% from 2025 to 2035
- The Worldwide Pediatric Non-invasive Wearables Market Size is Expected to Reach USD 10.80 Billion by 2035
- Asia Pacific is expected to grow the fastest during the forecast period.
Get more details on this report -
According to a research report published by Spherical Insights and Consulting, the global pediatric non-invasive wearables market size was worth around USD 2.75 Billion in 2024 and is predicted to grow to around USD 10.80 Billion by 2035 with a compound annual growth rate (CAGR) of 13.24% from 2025 and 2035. The market for pediatric non-invasive wearables has a number of opportunities to grow due to the inclination towards predictive, personalized, and proactive healthcare, especially for pediatric patients.
Market Overview
The global industry of pediatric non-invasive wearables encompasses child-friendly devices like smartwatches, patches, and sensors designed for monitoring vital signs such as heart rate, temperature, and sleep patterns without skin penetration. Pediatric non-invasive wearables refer to wearable sensors (including smartwatches, patches, and smart clothing) that monitor, track, and record children’s vital signs and physiological data in real-time without piercing the skin. There is growing automation in monitoring configurations, while specialized multi-parameter compatibility systems support multiple patient applications and operational condition compatibility.
Innovation and market expansion are anticipated as a result of major players' growing R&D expenditures and expanding partnerships. For instance, in February 2024, Sibel Health, a leading digital health company, announced that its continuous wearable monitoring solution, ANNE One, had received another 510(k) clearance from the U.S. Food and Drug Administration (FDA).
Report Coverage
This research report categorizes the pediatric non-invasive wearables market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the pediatric non-invasive wearables market. Recent market developments and competitive strategies such as expansion, type launch, development, partnership, merger, and acquisition have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the pediatric non-invasive wearables market.
Global Pediatric Non-Invasive Wearables Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 2.75 Billion |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | CAGR Of 13.24% |
| 2035 Value Projection: | USD 10.80 Billion |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 210 |
| Tables, Charts & Figures: | 112 |
| Segments covered: | By Monitoring Type,By Patient Age Group |
| Companies covered:: | By Monitoring Type,By Patient Age Group |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
The market for pediatric non-invasive wearables is driven by an increasing need for enhanced patient monitoring performance and advanced health data management initiatives. For instance, in November 2025, First Philips Azurion system was installed in an initiative to expand access to advanced care in Indonesia. An increasing prevalence of chronic diseases such as asthma and obesity, along with an increasing premature birth rate, is anticipated to drive the market demand for pediatric non-invasive wearables.
Restraining Factors
The pediatric non-invasive wearables market is restricted by factors like high device costs and technical complexities associated with the integration of specialized monitoring protocols into existing healthcare management processes.
Market Segmentation
The pediatric non-invasive wearables market share is classified into monitoring type and patient age group.
- The vitals and temperature wearables segment dominated the market with over 37.0% share in 2024 and is projected to grow at a substantial CAGR during the forecast period.
Based on the monitoring type, the pediatric non-invasive wearables market is divided into vitals and temperature wearables, SpO2 and respiratory wearables, activity and sleep wearables, and other. Among these, the vitals and temperature wearables segment dominated the market with over 37.0% share in 2024 and is projected to grow at a substantial CAGR during the forecast period. Vitals and temperature monitoring systems have superior precision control properties and operational efficiency characteristics. Temperature is proven to be an efficient infection-detection system with viruses like influenza, RSV, COVID-19, adenovirus, and enteroviruses, especially in at-home settings with an increasing demand for wearable monitoring devices.
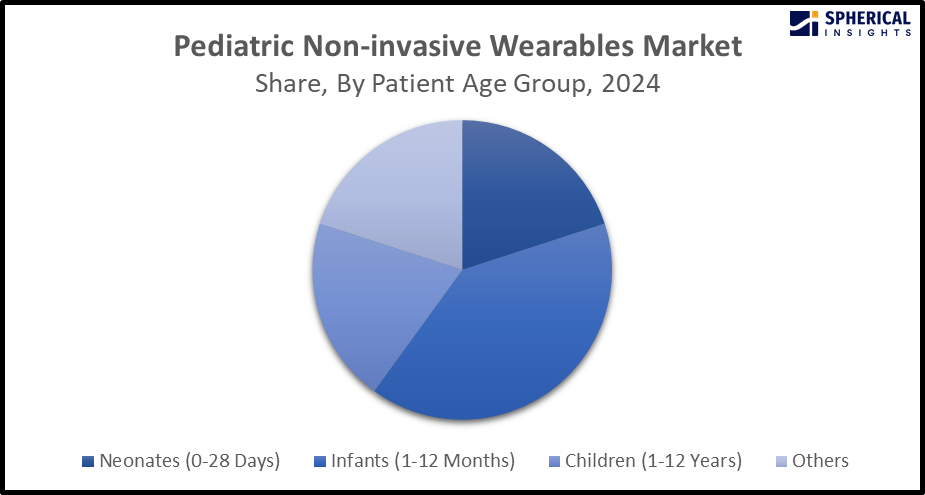
- The infants (1-12 years) segment accounted for the dominant market share of about 39.5% in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the patient age group, the pediatric non-invasive wearables market is divided into neonates (0-28 days), infants (1-12 months), children (1-12 years), and others. Among these, the infants (1-12 years) segment accounted for the dominant market share of about 39.5% in 2024 and is anticipated to grow at a significant CAGR during the forecast period. Infants' health monitoring is important, which must be continuously monitored for detecting abnormalities, and the data must be saved on a cloud server and retrieved through a particular mobile app, allowing caregivers to track the baby's health remotely. In order to support age-specific requirements and thorough operational control procedures, healthcare facilities are optimizing equipment selections.
Get more details on this report -
Regional Segment Analysis of the Pediatric Non-invasive Wearables Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the pediatric non-invasive wearables market over the predicted timeframe.
North America is anticipated to hold the largest share of approximately 39.5% in the pediatric non-invasive wearables market over the predicted timeframe. The market ecosystem in North America is strong, with both cutting-edge technologies like wearable and implantable BioMEMSs (biomedical microelectromechanical systems). The demand for pediatric non-invasive wearables has been driven by the region's increasing parental awareness about proactive, real-time health monitoring, along with the prevalence of childhood chronic diseases such as asthma, diabetes, and congestive heart diseases. The U.S. is dominating the North America pediatric non-invasive wearables market, with a major market share within the region, owing to the increased adoption of remote patient monitoring, advanced healthcare infrastructure, and the presence of industrial players.
Asia Pacific is expected to grow at a rapid CAGR of around 12.0% in the pediatric non-invasive wearables market during the forecast period. The Asia Pacific area has a thriving market for pediatric non-invasive wearables due to advancements in sensors, wireless connectivity, and real-time integration. Due to their governments' increasing partnership and upsurging initiatives plays vital role in propelling the market expansion. For instance, in November 2024, Murdoch Children’s Research Institute (MCRI) would partner with 12 countries in the Pacific and Southeast Asia under a $10 million initiative to improve child and adolescent health across the region. China is the leading country in the Asia Pacific pediatric non-invasive wearables market, driven by an increased adoption of smart healthcare, increasing obesity/diabetes rates, and a strong market position.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the pediatric non-invasive wearables market, along with a comparative evaluation primarily based on their type of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes type development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Philips
- Masimo
- Withings
- Fitbit
- Garmin
- Owlet
- Xiaomi
- Apple
- Samsung
- iRhythm
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting And Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Developments
• In June 2025, Sonu, an AI-enabled wearable device, was approved for use in pediatric patients aged 12 years and older to relieve moderate to severe nasal congestion caused by allergic and non-allergic rhinitis.
• In March 2025, the pediatric health system Connecticut Children’s expanded its partnership with Xerox by introducing an AI-powered solution aimed at streamlining care delivery while improving patient outcomes.
• In January 2025, CapsoVision, a leader in capsule endoscopy, announced that its flagship product, CapsoCam Plus, received FDA clearance for use in pediatric patients aged two years and above.
• In February 2024, Sibel Health, a leading digital health company, announced that its continuous wearable monitoring solution, ANNE One, received another 510(k) clearance from the U.S. Food and Drug Administration (FDA).
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the pediatric non-invasive wearables market based on the below-mentioned segments:
Global Pediatric Non-invasive Wearables Market, By Monitoring Type
- Vitals and Temperature Wearables
- SpO2 and Respiratory Wearables
- Activity and Sleep Wearables
- Other
Global Pediatric Non-invasive Wearables Market, By Patient Age Group
- Neonates (0-28 Days)
- Infants (1-12 Months)
- Children (1-12 Years)
- Others
Global Pediatric Non-invasive Wearables Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Need help to buy this report?