Japan Biosimilars Market Size, Share, By Product (Recombinant Human Growth Hormone (RHGH), Granulocyte Colony-Stimulating Factor, Interferon, and Others), By Application (Offsite Treatment, Oncology, Chronic Disorder, Autoimmune Disease, and Others), Japan Biosimilars Market Industry Trend, Forecasts to 2035
Industry: HealthcareJapan Biosimilars Market Insights Forecasts to 2035
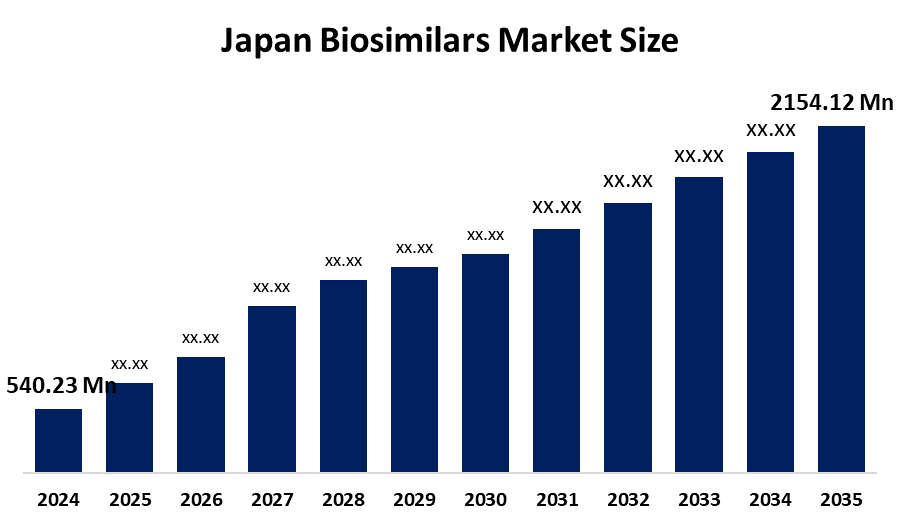
- Japan Biosimilars Market Size 2024: USD 540.23 million
- Japan Biosimilars Market Size 2035: USD 2154.12 million
- Japan Biosimilars Market CAGR 2024: 13.4%
- Japan Biosimilars Market Segments: Product and Application
Get more details on this report -
The Japan biosimilars market involves the production, sale, and use of medicines that are highly similar to approved biologic drugs but at lower costs. These medicines treat chronic diseases like cancer, diabetes, and autoimmune disorders. Hospitals, clinics, and pharmacies distribute them under strict regulatory oversight from the Ministry of Health, Labour and Welfare. Furthermore, the Japan Biosimilars Market is growing due to rising healthcare costs, patent expirations of original biologics, and increasing prevalence of chronic diseases. Hospitals and clinics are adopting biosimilars to reduce expenses. Improved manufacturing technologies and increased physician awareness support market expansion. National health insurance coverage and patient acceptance.
The Japan government has implemented biosimilar regulations through the Pharmaceuticals and Medical Devices Agency. The policies emphasise clinical trial tests and similarity tests to prove the safety and efficacy of the drugs. Pricing policies favour the use of biosimilars by hospitals. The national insurance policies cover biosimilars to promote the drugs while maintaining strict regulations to prevent misuse.
The Japan biosimilars market trends are the introduction of new-generation biosimilars with better delivery systems, increasing use of biosimilars in oncology and autoimmune diseases, and collaboration between biotech companies and hospitals. Japan is witnessing pharmacist-substitution programs led by pharmacists. Patient tracking systems and increasing physician trust are influencing biosimilar market trends in Japan, along with new emerging areas of treatment.
Japan Biosimilars Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2024 |
| Market Size in 2024: | USD 540.23 Million |
| Forecast Period: | 2025-2035 |
| Forecast Period CAGR 2025-2035 : | CAGR Of 13.4 % |
| 2035 Value Projection: | USD 2154.12 Million |
| Historical Data for: | 2020-2023 |
| No. of Pages: | 191 |
| Tables, Charts & Figures: | 102 |
| Segments covered: | By Product,By Application |
| Companies covered:: | Daiichi Sankyo Biosimilars, Nippon Kayaku Biosimilars, Fujifilm Kyowa Kirin Biologics, JCR Pharmaceuticals, Meiji Seika Pharma Co., Ltd., Nichi-Iko Pharmaceutical Co., Ltd., Mochida Pharmaceutical Co., Ltd., Sandoz International GmbH, Samsung Bioepis Co., Ltd., Celltrion Healthcare Co., Ltd., Pfizer Inc., and Others. |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Market Dynamics of the Japan Biosimilars Market:
The driving factors of the Japan biosimilars market are the increase in the rate of chronic diseases, patent expiration of biological drugs, financial constraints faced by the healthcare sector, and the increased acceptability of biosimilars among doctors as well as patients. Technological advancements in the production of biosimilars increase trust among consumers as well as doctors. Government incentives are also a driving force for hospitals to use biosimilars.
The Japan Biosimilars Market has certain restraints as well, which are mainly regulatory challenges, lengthy approval times, and costly development of biosimilars. Less awareness among doctors as well as patients is another restraint faced by the biosimilars market. Immunogenicity is another concern that acts as a restraint for the biosimilars market. Patent litigations are another restraint faced by the biosimilars market.
The Opportunities lie in developing biosimilars for cancer, autoimmune, and rare diseases, and expanding hospital and retail adoption. Cost-effective alternatives to expensive biologics can increase market penetration. Collaborations with local biotech firms and global companies can drive innovation. Japan’s ageing population and rising chronic disease burden create new patient segments. Digital health and personalised therapies further enhance market potential.
Market Segmentation
The Japan biosimilars market share is classified into products and applications
By Product:
The Japan biosimilars market is divided by product into recombinant human growth hormone, granulocyte colony-stimulating factor, interferon, and others. Among these, the granulocyte colony-stimulating factor segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. The granulocyte colony-stimulating factor segment dominates due to its widespread use in preventing chemotherapy-induced neutropenia, high physician trust, proven clinical effectiveness, strong hospital adoption, and Japan’s growing cancer patient population, making it the most prescribed and high-demand biosimilar compared to RHGH, interferons, and other biosimilar products.
By Application:
The Japan biosimilars market is divided by application into off-site treatment, oncology, chronic disorder, autoimmune disease, and others. Among these, the oncology segment dominated the share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. The oncology segment dominates due to Japan’s rising cancer prevalence, strong hospital and physician adoption, and proven effectiveness of biosimilars in chemotherapy support. High demand for cost-effective treatments, frequent hospital administration, and long-term patient care make oncology the largest application segment compared to off-site treatment, chronic disorders, autoimmune diseases, and other uses.
Competitive Analysis:
The report offers the appropriate analysis of the key organisations/companies involved within the Japan biosimilars market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborate analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the Japan Biosimilars Market:
• Daiichi Sankyo Biosimilars
• Nippon Kayaku Biosimilars
• Fujifilm Kyowa Kirin Biologics
• JCR Pharmaceuticals
• Meiji Seika Pharma Co., Ltd.
• Nichi-Iko Pharmaceutical Co., Ltd.
• Mochida Pharmaceutical Co., Ltd.
• Sandoz International GmbH
• Samsung Bioepis Co., Ltd.
• Celltrion Healthcare Co., Ltd.
• Pfizer Inc.
• Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the Japan, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the Japan biosimilars market based on the below-mentioned segments:
Japan Biosimilars Market, By Product.
- Recombinant Human Growth Hormone (RHGH)
- Granulocyte Colony-Stimulating Factor
- Interferon
- Others
Japan Biosimilars Market, By Application
- Offsite Treatment
- Oncology
- Chronic Disorder
- Autoimmune Disease
- Others
Need help to buy this report?