Global Gene Therapy Market Size By Vector Type (Viral Vector, Non-Viral Vector), By Therapy (In Vivo Therapy, Ex Vivo Therapy), By Gene Type (Antigen, Cytokine, Tumor Suppressor, Suicide, Deficiency, Growth factors, Receptors, Others), By Application (Oncological Disorders, Rare Diseases, Neurological Disorders, Other Diseases), By Geographic Scope And Forecast, 2022 - 2032
Industry: HealthcareGlobal Gene Therapy Market Insights Forecasts to 2032
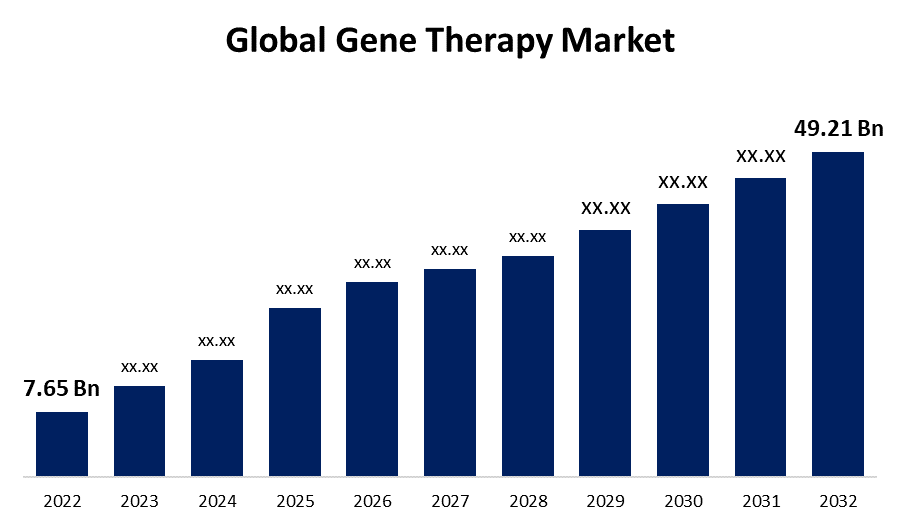
- The Global Gene Therapy Market Size was valued at USD 7.65 Billion in 2022.
- The Market Size is Growing at a CAGR of 20.46% from 2022 to 2032
- The Worldwide Gene Therapy Market Size is expected to reach USD 49.21 Billion by 2032
- Asia Pacific Market Size is expected to Grow the fastest during the forecast period
Get more details on this report -
The Global Gene Therapy Market Size is expected to reach USD 49.21 Billion by 2032, at a CAGR of 20.46% during the forecast period 2022 to 2032.
Gene therapy involves the introduction, alteration, or replacement of genetic material to treat or prevent various diseases, including inherited genetic disorders, certain types of cancer, and rare conditions. This field holds immense promise for revolutionizing healthcare by addressing the root causes of diseases at a genetic level. In recent years, the gene therapy market has witnessed remarkable growth due to advancements in biotechnology, increased understanding of genomics, and a surge in research and development activities. The market's expansion is driven by a growing demand for innovative and curative treatments, especially for previously untreatable or poorly managed genetic diseases. Key factors influencing the gene therapy market include regulatory approvals, technological advancements, funding for research, and the involvement of pharmaceutical and biotech companies. While gene therapy offers great potential, it also faces challenges related to safety, efficacy, and affordability, which necessitate ongoing research and development efforts and close collaboration with regulatory agencies.
Market Outlook
Gene Therapy Market Price Analysis
Price analysis in the gene therapy market is influenced by several factors. One key consideration is the complexity of the treatment, which often requires highly specialized techniques and tools, impacting overall costs. Additionally, factors such as research and development expenses, manufacturing costs, clinical trials, and regulatory requirements contribute to pricing dynamics. As gene therapy continues to evolve and gain traction, striking a balance between affordability and ensuring access to groundbreaking treatments remains a critical challenge for healthcare stakeholders worldwide.
Gene Therapy Market Distribution Analysis
The global gene therapy market's distribution analysis plays a vital role in ensuring that innovative genetic treatments reach the patients who need them. Distribution in this market involves various elements, including the geographic spread of treatment centers, the logistics of delivering gene therapy products, and the involvement of pharmaceutical companies and healthcare providers. The availability and accessibility of gene therapies can vary significantly from one region to another, influenced by regulatory approvals, healthcare infrastructure, and reimbursement policies. As the field continues to expand, analyzing and optimizing distribution channels becomes crucial to ensure that cutting-edge gene therapies can effectively reach the global patient population in need.
Global Gene Therapy Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2022 |
| Market Size in 2022: | USD 7.65 Billion |
| Historical Data for: | 2019-2021 |
| No. of Pages: | 200 |
| Tables, Charts & Figures: | 110 |
| Segments covered: | By Vector, By Therapy, By Gene Type, By Geographic Scope |
| Companies covered:: | REGENXBIO, Inc., Oxford BioMedica plc, Dimension Therapeutics, Inc., Bristol-Myers Squibb Company, SANOFI, Applied Genetic Technologies Corporation, F. Hoffmann-La Roche Ltd, bluebird Bio, Inc., Novartis AG, Taxus Cardium Pharmaceuticals Group, Inc. (Gene Biotherapeutics), UniQure N.V., Shire Plc, Cellectis S.A., Sangamo Therapeutics, Inc, and Other Key Players. |
| Growth Drivers: | Increasing the number of regulatory approvals for gene therapy products |
| Pitfalls & Challenges: | COVID-19 Empact,Challenges, Future, Growth, & Analysis |
Get more details on this report -
Market Dynamics
Gene Therapy Market Dynamics
Increasing the number of regulatory approvals for gene therapy products
Before being approved, gene therapy products are subjected to extensive regulatory scrutiny. They act at the genetic level to produce therapeutic effects that are more effective and long-lasting than traditional medicines. Because of the significant advantages offered by these products, the number of approvals for gene therapy products has increased. Between 2021 and 2023, the US FDA approved more than ten products. Furthermore, a substantial number of products in the clinical pipeline are expected to receive final approval during the forecast period. As a result, this is a major factor driving market growth during the forecast period.
Restraints & Challenges
Gene therapy products are expensive
Research and development efforts for advanced therapies such as gene therapy are extensive. As a result, they necessitate a significant investment of both time and money. Laboratory research, preclinical testing, clinical trials, and regulatory approval are all included in the costs. Furthermore, vectors are used in gene therapy products, which must be produced and separately engineered as needed. This adds to the overall complexity of the manufacturing process. Manufacturing processes must be tightly controlled, and strict quality standards must be met, which raises production costs. Furthermore, gene therapies are highly targeted therapies that are not mass produced. All of these factors contribute to the high costs of gene therapy products, which limit their adoption, particularly in developing regions around the world.
Regional Forecasts
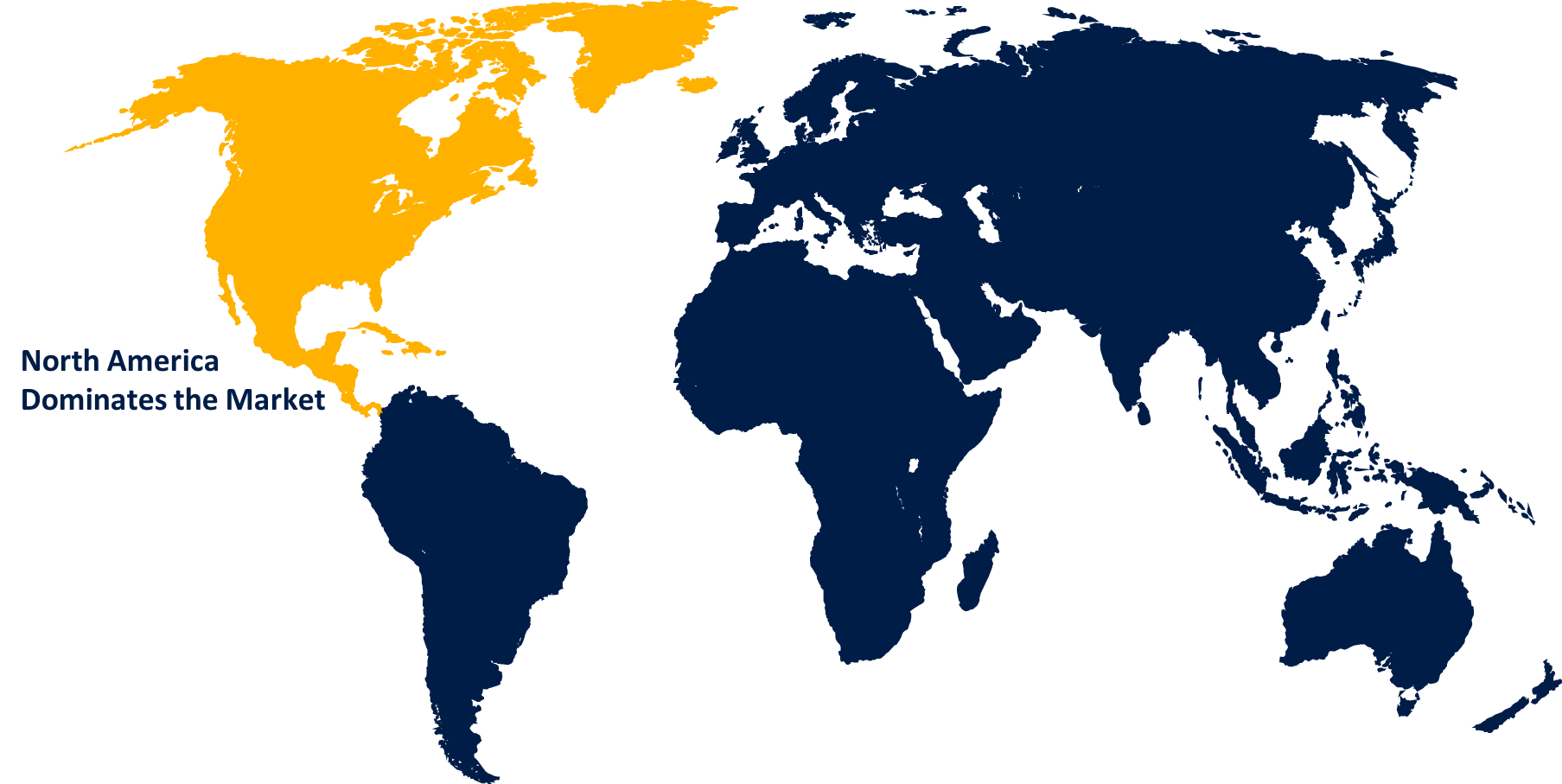
North America Market Statistics
Get more details on this report -
North America is anticipated to dominate the Gene Therapy Market from 2023 to 2032. During the forecast period, this region is projected to become the largest routine manufacturer of gene therapy in terms of approvals and revenue generated. Increased R&D investments from large and small companies in the development of ideal therapy drugs are expected to drive the market even further.
Asia Pacific Market Statistics
Asia Pacific is witnessing the fastest market growth between 2023 to 2032. This is due to the country's large population, which has unmet medical needs, as well as the growing demand for novel technologies in the treatment of rare but increasingly prevalent diseases. The Asia Pacific market for commercial application of genetic therapies is expected to grow significantly during the forecast period, owing to the easy availability of resources, the local presence of major companies, and increased government investment.
Segmentation Analysis
Insights by Therapy
The in vivo therapy segment accounted for the largest market share over the forecast period 2023 to 2032. In vivo gene therapy had a dominant market share of around 65.3% in 2022 and is projected to grow rapidly during the forecast period. Systemic treatment is possible with in vivo gene therapy because it can reach multiple sites or organs throughout the body. This is especially useful for diseases that affect multiple organ systems or have systemic manifestations, as it allows for a more comprehensive therapeutic approach.
Insights by Vector Type
The non viral vector segment accounted for the largest market share over the forecast period 2023 to 2032. The rise in the incidence of cancer and other target diseases, the availability of reimbursement policies, and the increase in funding for gene therapy research are the key factors anticipating market growth in the coming years.
Recent Market Developments
- In May 2022, Biogen Inc. (US) and Scribe Therapeutics (US) have collaborated to develop a new neurological disease target in gene therapy using the company's CRISPR technology. This partnership had a deal worth USD 15 million.
Competitive Landscape
Major players in the market
- REGENXBIO, Inc.
- Oxford BioMedica plc
- Dimension Therapeutics, Inc.
- Bristol-Myers Squibb Company
- SANOFI
- Applied Genetic Technologies Corporation
- F. Hoffmann-La Roche Ltd
- bluebird Bio, Inc.
- Novartis AG
- Taxus Cardium Pharmaceuticals Group, Inc. (Gene Biotherapeutics)
- UniQure N.V.
- Shire Plc
- Cellectis S.A.
- Sangamo Therapeutics, Inc
- Others
Market Segmentation
This study forecasts revenue at global, regional, and country levels from 2023 to 2032.
Gene Therapy Market, Vector Type Analysis
- Viral Vector
- Non Viral Vector
Gene Therapy Market, Therapy Analysis
- In Vivo Therapy
- Ex Vivo Therapy
Gene Therapy Market, Gene Type Analysis
- Antigen
- Cytokine
- Tumor Suppressor
- Suicide
- Deficiency
- Growth factors
- Receptors
- Others
Gene Therapy Market, Application Analysis
- Oncological Disorders
- Rare Diseases
- Neurological Disorders
- Other Diseases
Gene Therapy Market, Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- Uk
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Need help to buy this report?