Asia-Pacific Multi-Cancer Early Detection Market Size, Share, and COVID-19 Impact Analysis, By Type (Liquid Biopsy, Gene Panel, LDT, & Others), By End-Use (Hospitals & Clinics, Diagnostics Laboratories, Others), By Region (India, China, Japan, and Rest of Asia-Pacific), and Asia-Pacific Multi-Cancer Early Detection Market Insights Forecasts 2023 – 2033
Industry: HealthcareAsia-Pacific Multi-Cancer Early Detection Market Insights Forecasts to 2033
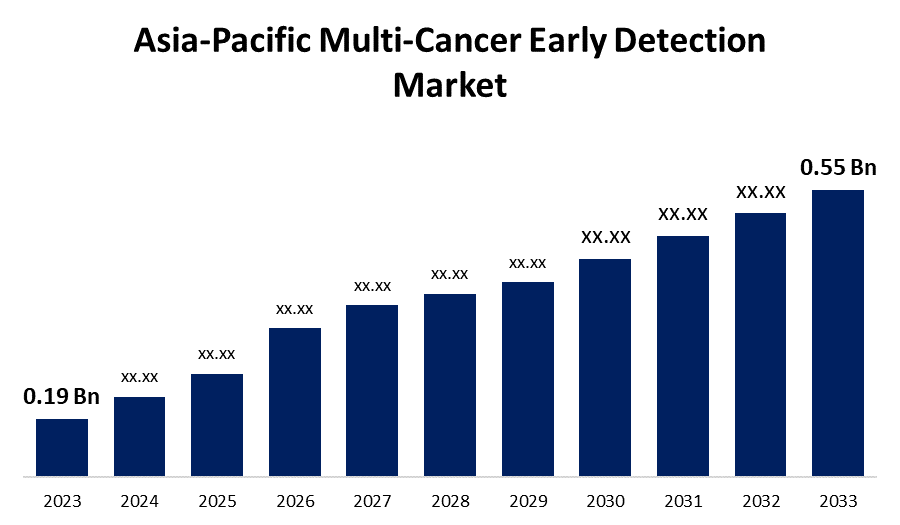
- The Asia-Pacific Multi-Cancer Early Detection Market Size was valued at USD 0.19 Billion in 2023.
- The Market Size is Growing at a CAGR of 11.21% from 2023 to 2033.
- The Asia-Pacific Multi-Cancer Early Detection Market Size Expected to Reach USD 0.55 Billion by 2033.
Get more details on this report -
The Asia-Pacific Multi-Cancer Early Detection Market Size is expected to reach USD 0.55 Billion by 2033, at a CAGR of 11.21% during the forecast period 2023 to 2033.
Market Overview
Multi-cancer early detection is a diagnostic test used to detect different types of cancer in the human body. The multi-cancer early detection test uses a variety of samples, including blood, liquid biopsy, gene, LTD, and others. This diagnostic test allows physicians to assess the risk of cancer cell multiplication inside a blood vessel, epidermis, colon, lungs, throat, and other body parts. In response, MCED is designed to detect tumors in the body. The multi-cancer early detection (MCED) market is expanding due to increased healthcare reforms, improved healthcare infrastructure, a growing population, and an increasing number of local companies entering the market. Thus, the use of cancer screening tests has increased in recent years as a result of growing government initiatives, such as free screening for breast cancer, lung cancer, and cervical cancer, as well as increased collaborations between research institutes, companies, and the government to distribute and supply these tests. Cancer has received increased attention in Asia Pacific due to demographic shifts caused by urbanization, aging populations, and economic expansion. A high prevalence of health risk factors, such as increased alcohol and cigarette consumption, poor diets, and sedentary lifestyles, are all contributing to an increase in cancer incidence. The rapidly increasing cancer burden may exceed the current capacity of healthcare systems in several Asia Pacific countries.
Report Coverage
This research report categorizes the market for the Asia-Pacific multi-cancer early detection market based on various segments and regions forecasts revenue growth and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the Asia-Pacific multi-cancer early detection market. Recent market developments and competitive strategies such as expansion, product launch, and development, partnership, merger, and acquisition have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the Asia-Pacific multi-cancer early detection market.
Asia-Pacific Multi-Cancer Early Detection Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2023 |
| Market Size in 2023: | USD 0.19 Billion |
| Forecast Period: | 2023-2033 |
| Forecast Period CAGR 2023-2033 : | 11.21% |
| 2033 Value Projection: | USD 0.55 Billion |
| Historical Data for: | 2019-2022 |
| No. of Pages: | 200 |
| Tables, Charts & Figures: | 110 |
| Segments covered: | By Type, By End-Use, By Region and COVID-19 Impact Analysis. |
| Companies covered:: | Grail, LLC (Illumina, Inc.), Exact Sciences Corporation, AnchorDx, Guardant Health, Burning Rock Biotech Limited, GENECAST, Singlera Genomics Inc., Laboratory for Advanced Medicine, Inc., MiRXES Pte Ltd., Lucence Health, Inc. and Other Key Vendors. |
| Pitfalls & Challenges: | COVID-19 Empact, Challenge, Future, Growth, & Analysis |
Get more details on this report -
Driving Factors
Rising cancer incidence is a key factor driving the growth of the Asia Pacific multi-cancer early detection market, as is the Asia-Pacific’s aging population. Furthermore, the Asia Pacific multi-cancer early detection market is expanding rapidly, driven in part by increased public awareness of the importance of cancer screening and early detection. Elevated cancer awareness has emerged as a powerful market driver, through a variety of initiatives, education campaigns, and the region's changing healthcare landscape. Furthermore, media coverage and the spread of information via digital platforms have helped to raise awareness of cancer risks and the importance of early diagnosis. Stories of survival and the most recent advancements in cancer diagnostics and treatment are now widely available to the public, empowering people to seek timely healthcare interventions.
Restraining Factors
The Asia Pacific multi-cancer early detection market faces an important obstacle in the form of the high cost of advanced diagnostics. Limited access to healthcare services hampers the growth of the Asia Pacific multi-cancer early detection market. The shortage of skilled healthcare professionals, especially in remote areas, exacerbates the situation. The collection of sufficient efficacy and safety data for regulatory approval is expected to hamper market growth.
Market Segment
- In 2023, the liquid biopsy segment accounted for the largest revenue share over the forecast period.
Based on type, the Asia-Pacific multi-cancer early detection market is segmented into liquid biopsy, gene panel, LDT, & others. Among these, the liquid biopsy segment has the largest revenue share over the forecast period. Liquid biopsies are minimally invasive procedures that involve the examination of bodily fluids like blood, urine, and saliva. Their non-invasive nature makes them appealing to patients who may be hesitant to undergo traditional invasive procedures such as tissue biopsies. It improves patient compliance and participation in early cancer screening programs. Liquid biopsy technology has received extensive research and investment from both the public and private sectors. Continuous improvements have increased the sensitivity and specificity of these tests. This progress has contributed to increased adoption and demand in the Asia Pacific multi-cancer early detection market.
- In 2023, the diagnostics laboratories segment is witnessing the largest growth over the forecast period.
Based on end use, the Asia-Pacific multi-cancer early detection market is segmented into hospitals & clinics, diagnostics laboratories, and others. Among these, the diagnostics laboratories segment is witnessing the largest growth over the forecast period. Diagnostic laboratories have specialized personnel, such as pathologists, clinical laboratory scientists, and technicians, who can perform complex cancer screening and early detection tests. This expertise is essential for producing accurate and reliable results. Diagnostic laboratories follow strict quality control procedures to ensure the reliability and consistency of test results. This is especially important for early cancer detection, as accurate results are required for timely intervention and treatment.
- The Japan is projected to have the largest share of the Asia-Pacific multi-cancer early detection market over the forecast period.
Based on region, the Japan is projected to have the largest share of the Asia-Pacific multi-cancer early detection market over the forecast period. Japan has one of the largest and fastest-aging populations, with a sizable proportion of elderly people. As people age, their chances of developing cancer increase, making early detection critical. This demographic factor has increased the demand for cancer screening services. Japan has a relatively high cancer incidence, including common cancer types like gastric cancer, lung cancer, and colorectal cancer. A Japan with high cancer rates emphasizes the importance of effective early detection.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the Asia-Pacific multi-cancer early detection market along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Grail, LLC (Illumina, Inc.)
- Exact Sciences Corporation
- AnchorDx
- Guardant Health
- Burning Rock Biotech Limited
- GENECAST
- Singlera Genomics Inc.
- Laboratory for Advanced Medicine, Inc.
- MiRXES Pte Ltd.
- Lucence Health, Inc.
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In May 2022, Guardant Health introduced "Shield," a Laboratory-Developed Test (LDT) blood assay intended for the early detection of colorectal cancer (CRC) in adults aged 45 and up. The test had an 83% sensitivity in identifying people with CRC while maintaining a consistent 90% specificity for those without advanced neoplasia and those who had negative colonoscopy results.
This study forecasts revenue at regional, and country levels from 2022 to 2033. Spherical Insights has segmented the Asia-Pacific Multi-Cancer Early Detection market based on the below-mentioned segments:
Asia-Pacific Multi-Cancer Early Detection Market, By Type
- Liquid Biopsy
- Gene Panel
- LDT
- Others
Asia-Pacific Multi-Cancer Early Detection Market, By End-Use
- Hospitals & Clinics
- Diagnostics Laboratories
- Others
Asia-Pacific Multi-Cancer Early Detection Market, By Region
- India, China
- Japan
- Rest of Asia-Pacific
Need help to buy this report?