Global Medical Device Testing Service Market Size To Worth USD 16.17 Billion By 2030 | CAGR of 9.3%
Category: HealthcareGlobal Medical Device Testing Service Market Size To Worth USD 16.17 Billion By 2030
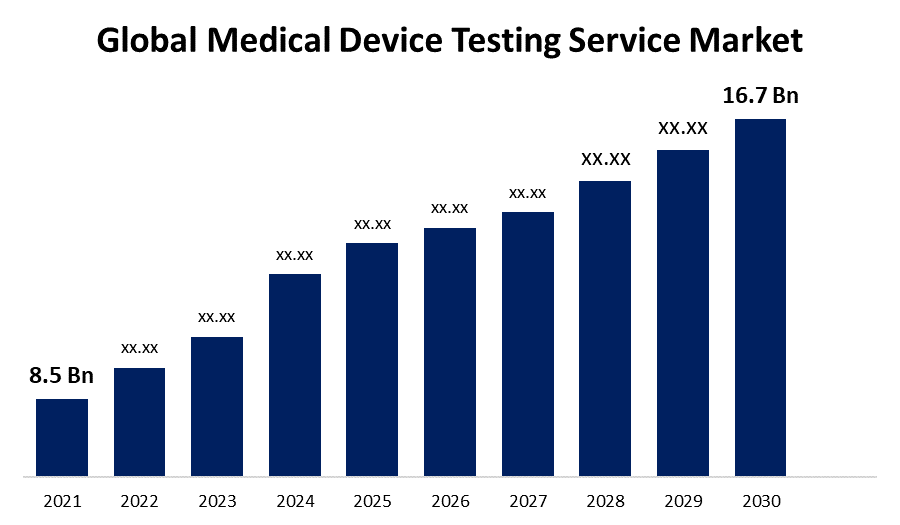
According to a research report published by Spherical Insights & Consulting, the Global Medical Device Testing Service Market Size to grow from USD 8.5 billion in 2021 to USD 16.17 billion by 2030, at a Compound Annual Growth Rate (CAGR) of 9.3% during the forecast period.
Get more details on this report -
Browse key industry insights spread across 210 pages with 100 market data tables and figures & charts from the report on "Global Medical Device Testing Service Market Size, Share, and COVID-19 Impact Analysis, By Service (Biocompatibility Tests, Chemistry Test, Microbiology & Sterility Testing, Package Validation), By Phase (Preclinical, Clinical), by Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2021 – 2030". Get Detailed Report Description Here: https://www.sphericalinsights.com/reports/medical-device-testing-service-market
Medical device testing is the procedure used to demonstrate a medical equipment's dependability and safety while being used. In the creation of new products, extensive design validation testing is utilised. This includes testing for toxicity, chemical analysis, and performance, as well as occasionally testing that involves humans or even clinical trials. Less resources are often required for continuous quality assurance testing. Typically included are dimensional checks, a few functional tests, and package verification. Medical testing services come in a wide range of varieties, including inspection and certification services.
COVID 19 Impact
With the start of the COVID 19 pandemic, governments of various nations have put in place various measures to stop the spread of the deadly virus, such as lockdown measures and limiting trade between various nations, which has disrupted the supply chain and had an effect on the global market for medical device testing services. Additionally, the global shortage of essential medical devices has been brought on by the interruption of the supply chain. As a result, different nations have taken action to address these shortages by importing machinery or domestically producing medical gadgets. In order to guarantee both market and product stability, it is also envisaged that domestic manufacturing of crucial medical devices will overcome trade obstacles.
Verification and validation procedures are widely and extensively employed in the healthcare industry. Generally speaking, validation examines whether usability standards have been met by a product after it has been utilised for its intended purpose. On the other hand, verification establishes whether a product has been created in accordance with the specifications. The most prevalent methods of verification and validation for medical devices include design, process, and software. Medical devices are also getting bigger and more complicated, and they occasionally use high-end, produced polymers. This makes validation and verification (V&V) even more important. The end outcomes include improved repeatability, fewer mistakes, a reduced need for rework and redesign, a speedier time to market, better competitiveness, and cheaper production costs.
The expansion of the growth of the global market for medical device testing will be hampered by the high cost of medical devices and the obstacles to local medical device development in some regions. In addition, the market developments have been hampered by the growing competition in the medical technology sector and the protracted foreign qualification process.
Services Insights
Microbiology & Sterility testing segment holds the highest market share over the forecast period
On the basis of services, the global medical device testing market is segmented into Biocompatibility Tests, Chemistry Test, Microbiology & Sterility Testing, Package Validation. Among these, the microbiology and sterility testing segment holds the highest market share over the forecast period. To remove or lower the danger of contamination in the production process that could have resulted in illnesses in users or patients, microbiology and sterility testing are performed. If such examinations are not conducted, the regulatory process for the devices may be delayed.
Phase Insights
Clinical segment is dominating the market with the highest market share over the forecast period
Based on the phase, the global medical device testing market is segmented into preclinical and clinical. Among these, the clinical segment is dominating the market with the highest market share over the forecast period. One of the key reasons determining the biggest market in the clinical phase is the cost of the medical device testing services, which is considerably greater than the price of pre clinical devices. Another important aspect that is influencing the segmental expansion is the fact that testing of medical devices requires more time in each step than it does in the preclinical stage and is also more intensive.
Regional Insights
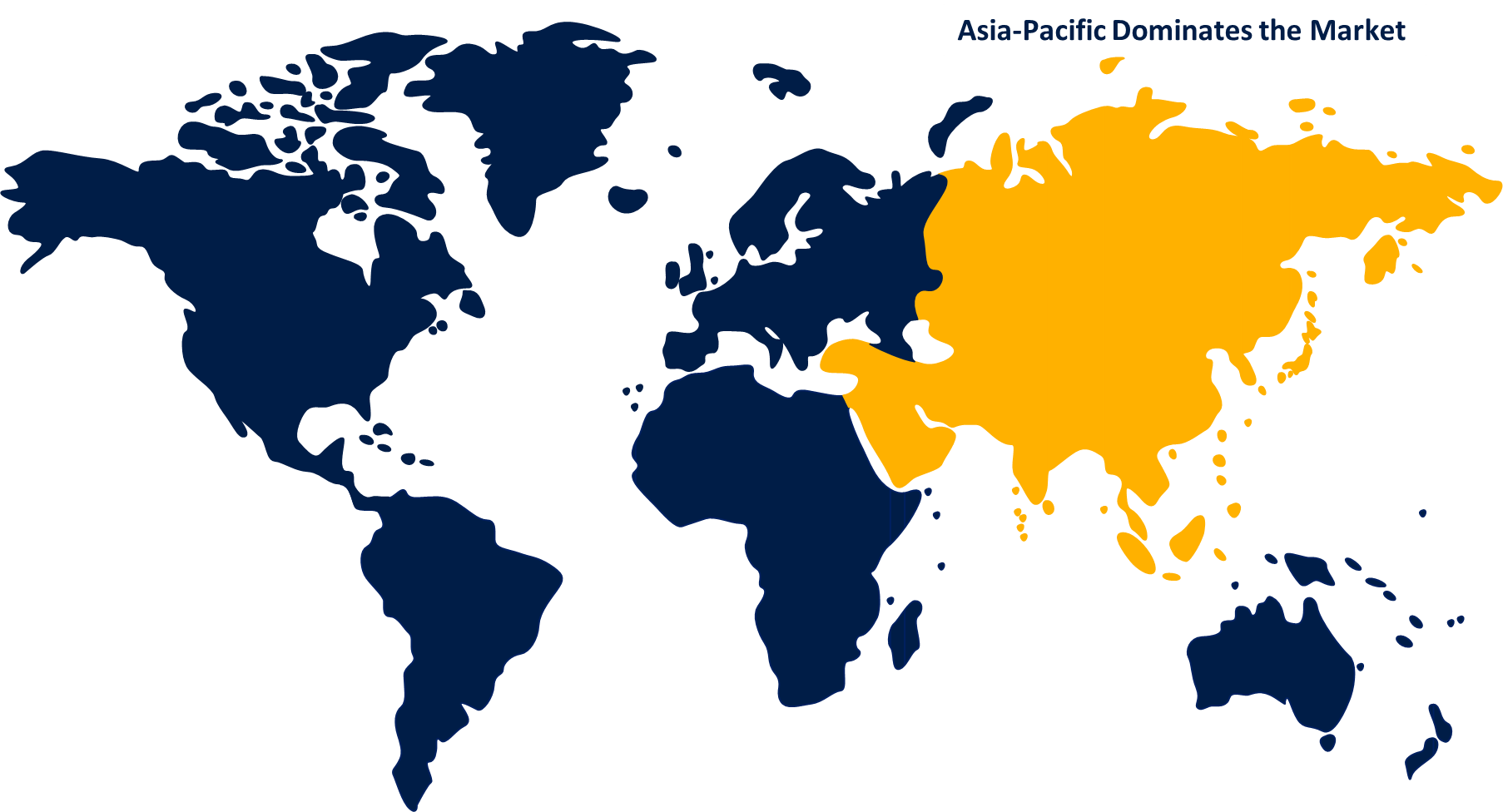
Asia Pacific is dominating the market with the largest market share over the forecast period
Get more details on this report -
The market is being dominated by Asia Pacific, which will have the greatest market share during the forecast. Due to China's tight product approval standards and the advancement of the healthcare infrastructure, there has been an increase in global interest in both the Chinese and Indian markets. Numerous product launches are taking place to maintain compliance with international standards because China has a significant position on the market for testing medical equipment.
On the other hand, North America is expected to see the fastest market growth during the projected period as a result of the complexity of product design and growing efforts to save costs. In addition, the existence of strong regulatory organisations like the FDA is fueling the expansion of the regional market. The region's need is expected to increase due to the increased production of medical devices to fulfil the region's high demand for effective healthcare.
Major vendors in the Global Medical Device Testing Market include SGS SA; Euro fins Scientific; Pace Analytical Services LLC; Intertek Group Plc; WUXI APPTEC; TÜV SÜD AG; Sterigenics International LLC; American Preclinical Services; North American Science Associates, Inc.; Charles River Laboratories International, Inc; Medical Device Testing Services; Toxikon, Inc.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2019 to 2030. Spherical Insights has segmented the global Medical Device Testing Service Market based on the below-mentioned segments:
Medical Device Testing Service Market, System Analysis
- Biocompatibility Tests
- Chemistry Test
- Microbiology & Sterility Testing
- Package Validation
Medical Device Testing Service Market, Phase Analysis
- Clinical
- Pre-clinical
Medical Device Testing Service Market, Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- Uk
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of Middle East & Africa
About the Spherical Insights & Consulting
Spherical Insights & Consulting is a market research and consulting firm which provides actionable market research study, quantitative forecasting and trends analysis provides forward-looking insight especially designed for decision makers and aids ROI.
Which is catering to different industry such as financial sectors, industrial sectors, government organizations, universities, non-profits and corporations. The company's mission is to work with businesses to achieve business objectives and maintain strategic improvements.
CONTACT US:
For More Information on Your Target Market, Please Contact Us Below:
Phone: +1 303 800 4326 (the U.S.)
Phone: +91 90289 24100 (APAC)
Email: inquiry@sphericalinsights.com, sales@sphericalinsights.com
Contact Us: https://www.sphericalinsights.com/contact-us
Follow Us: LinkedIn | Facebook | Twitter
Need help to buy this report?