
Top 20 Companies in Global Regulatory Affairs Outsourcing Market (2025–2035): Expert View by Spherical Insights
RELEASE DATE: Apr 2026 Author: Spherical InsightsRequest Free Sample Speak to Analyst
Introduction
The regulatory affairs outsourcing is a process by which companies outsource the process of ensuring the approval of drugs, biologics, and devices by third-party companies. In the context of the global regulatory affairs outsourcing market, the application of this process includes product registration and clinical trial submissions, regulatory writing and publishing (such as CTD dossier writing), strategic consulting, post-marketing surveillance and lifecycle maintenance, and legal representation for ensuring compliance with regional regulatory requirements. The global regulatory affairs outsourcing market is changing through the use of AI-based automation technologies, which are improving the accuracy of documentation and reducing submission timelines. The use of advanced analytics and real-world evidence is also improving regulatory decision-making. Long-term strategic relationships are replacing project-based relationships. The opportunities for the future are related to the growth in personalized medicine and biopharmaceuticals, which will require specialized regulatory expertise, as well as growth in global clinical trials and data-based compliance.
The China National Medical Products Administration enforces stringent, evolving compliance frameworks, increasing approval timelines by 20% and regulatory costs by 15%, accelerating demand in the global Regulatory Affairs Outsourcing Market.
Navigate Future Markets with Confidence: Insights from Spherical Insights LLP
The insights presented in this blog are derived from comprehensive market research conducted by Spherical Insights LLP, a trusted advisory partner to leading global enterprises. Backed by in-depth data analysis, expert forecasting, and industry-specific intelligence, our reports empower decision-makers to identify strategic growth opportunities in fast-evolving sectors. Clients seeking detailed market segmentation, competitive landscapes, regional outlooks, and future investment trends will find immense value in the full report. By leveraging our research, businesses can make informed decisions, gain a competitive edge, and stay ahead in the transition toward sustainable and profitable solutions.
Unlock exclusive market insights—Download the Brochure now and dive deeper into the future of the Global Regulatory Affairs Outsourcing Market .
Regulatory Affairs Outsourcing Market Size & Statistics
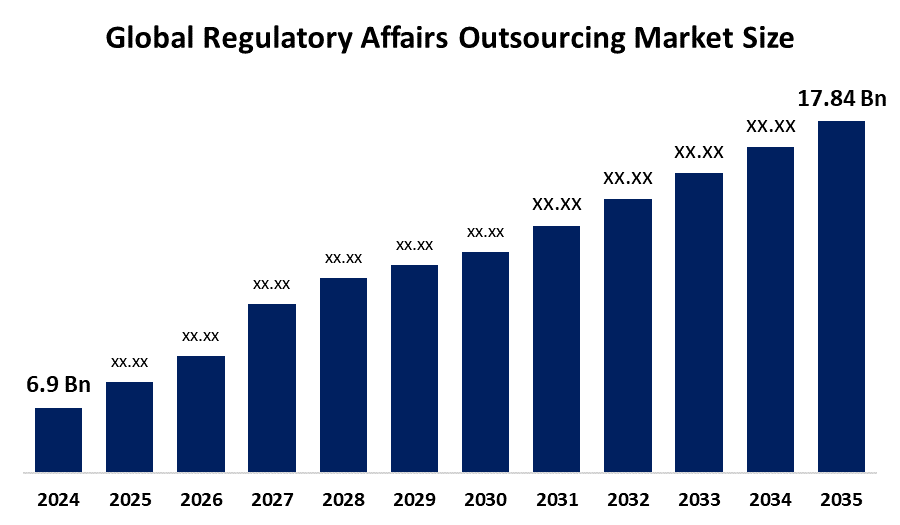
- The Market Size for Regulatory Affairs Outsourcing Market Was Estimated to be worth USD 6.9 billion in 2024.
- The Market Size is Going to Expand at a CAGR of 9.02% between 2025 and 2035.
- The Global Regulatory Affairs Outsourcing Market Size is anticipated to reach USD 17.84 billion by 2035.
- North America is estimated to generate the highest demand during the forecast period in the Global Regulatory Affairs Outsourcing Market.
- Asia-Pacific is projected to grow the fastest during the forecast period in The Global Regulatory Affairs Outsourcing Market.
Market Segmentation
Global Regulatory Affairs Outsourcing Market Size, Share, By Service Type (Regulatory Consulting, Legal Representation), By Size (Small, Medium), By Category Type (Drugs, Generics), By Indication (Oncology, Neurology), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 - 2035
Region al growth and demand
Asia-Pacific is expected to grow the fastest during the forecast period in the Regulatory Affairs Outsourcing Market. It is due to the expanding pharmaceutical and biotechnology industries, increasing clinical trial activities, and cost-effective outsourcing destinations such as India and China. Additionally, evolving regulatory frameworks and rising demand for faster product approvals are encouraging companies to outsource regulatory functions, supporting significant regional market expansion.
Unlock exclusive market insights—Download the Brochure now and dive deeper into the future of the Global Regulatory Affairs Outsourcing Market .
The Japan Government policies promoting generic drug adoption to reduce healthcare costs are increasing regulatory approval requirements, driving demand for regulatory consulting and outsourcing services in the global market.
North America is anticipated to generate the highest demand during the forecast period in the Regulatory Affairs Outsourcing Market. It is because of the strong presence of pharmaceutical companies, high investment in research and development, and strict regulatory environments maintained by regulatory bodies such as the FDA. The increasing complexity of drug approvals and regulations is prompting companies to outsource their regulatory needs, thus increasing the market demand.
The U.S. FDA expedited pathways such as Fast Track and Breakthrough Therapy are increasing regulatory complexity and timelines, driving demand for specialized outsourcing services in the global regulatory affairs outsourcing market.
Ready to lead the Regulatory Affairs Outsourcing Market
Discover the regional trends and growth factors shaping the industry. We’re here to assist with expert, personalized data.
Call +1 303 800 4326 or Send us a message for a personalized consultation.
Top 10 trends in the Regulatory Affairs Outsourcing Market
- AI-Driven Automation & RegTech
- Shift to Outcome-Based Outsourcing Models
- Growing Demand in Emerging Markets
- Strategic Focus on Post-Market Surveillance
- Rise of Functional Service Partnerships (FSP)
- Need for Advanced Therapy Expertise
- Increased Focus on Data Security & Quality
- Consolidation and M&A Activity
- Integration of Real-World Evidence (RWE)
- Nearshoring and Regional Hub Specialization
Unlock exclusive market insights—Download the Brochure now and dive deeper into the future of the Global Regulatory Affairs Outsourcing Market .
1.AI-Driven Automation & RegTech
In order to increase efficiency and decrease human error, outsourcing companies are using artificial intelligence, machine learning, and regulatory technology (RegTech) to automate repetitive operations including document validation, data extraction, and dossier preparation.
2.Shift to Outcome-Based Outsourcing Models
The Contracts are shifting from transactional, time-and-material models to outcome-based models, in which suppliers are compensated according to performance indicators like quicker submission times or successful product acceptance.
3.Growing Demand in Emerging Markets
It is because pharma and biotech businesses are boosting outsourcing to Asia-Pacific and Latin America to handle local regulatory challenges, leverage cheaper labor costs, and access specialized expertise for global expansion.
4.Strategic Focus on Post-Market Surveillance
The companies are increasingly outsourcing pharmacovigilance, lifecycle management, and post-approval maintenance due to stricter medication safety standards. This is a rapidly expanding market niche.
5.Rise of Functional Service Partnerships (FSP)
The businesses are choosing FSP models, which enable them to lease specialized regulatory personnel from CROs for certain projects rather than outsourcing entire departments, in order to preserve control while scaling.
Unlock exclusive market insights—Download the Brochure now and dive deeper into the future of the Global Regulatory Affairs Outsourcing Market .
Top 20 Companies Leading the Regulatory Affairs Outsourcing Market
- IQVIA
- ICON plc
- Thermo Fisher Scientific (PPD)
- Labcorp Drug Development
- Syneos Health
- Parexel International
- Charles River Laboratories
- Freyr Solutions
- ProPharma Group
- Celegence
- Facet Life Sciences
- CCS Associates
- Global Pharma Solutions
- Bentley Biomedical Consulting
- Veristat
- BlueReg
- CliniExperts
- Lambda Therapeutic Research
- Premier Research
- TFS HealthScience
1.IQVIA
Headquarters: Mumbai, India
The IQVIA, which was founded in 1982 and has 93,000 professionals worldwide, reported revenue of USD 16.31 billion in 2025. The firm specializes in providing integrated regulatory affairs outsourcing services, which include clinical trial management, regulatory submissions, and compliance, in order to help pharmaceutical and biotech organizations speed up their product approvals in the global regulatory affairs outsourcing market.
2. ICON plc
Headquarters: Dublin, Ireland
The ICON plc is a company that was founded in 1990 and has around 40,000-41,900 professionals worldwide. The company generated an approximate revenue of around 8.1-8.3 billion dollars in 2025. The company offers outsourced regulatory affairs, clinical development, and commercialization services that help pharmaceutical and biotechnology companies to increase operational efficiencies in the global regulatory affairs outsourcing industry.
3. Thermo Fisher Scientific (PPD)
Headquarters: Waltham, Massachusetts, United States
The Founded in 1956 and rebranded as Thermo Fisher Scientific (PPD), with PPD founded in 1985 and acquired in 2021. The firm has around 125,000 professionals worldwide. The firm reported revenue of around 44.56 billion USD in 2025. The firm provides regulatory affairs outsourcing, clinical development, and laboratory services through its clinical research division, PPD, for efficient regulatory compliance and quick drug approvals in the global regulatory affairs outsourcing market.
4. Syneos Health
Headquarters: Morrisville, North Carolina, United States
The Syneos Health, which was formed in 2017 following the merger of INC Research and inVentiv Health, is a global company with around 28,000 professionals. The company’s estimated revenue for 2025 is around 5.5-5.7 billion USD. Syneos Health provides regulatory affairs outsourcing services, clinical development services, and commercialization services, which allow biopharmaceutical companies to improve time-to-market efficiency in the global regulatory affairs outsourcing services market.
5. Labcorp Drug Development
Headquarters: Burlington, North Carolina, United States
The Labcorp Drug Development is the division of the Laboratory Corporation of America Holdings. The parent company was founded in 1978, and the company has around 70,000 professionals worldwide. The company generated around 13.6 billion USD in revenue in 2025. Labcorp Drug Development offers regulatory affairs outsourcing, clinical trial management, and lab services, which helps the pharmaceutical and biotech industry speed up their approval process and stay compliant in the global regulatory affairs outsourcing market.
Unlock exclusive market insights—Download the Brochure now and dive deeper into the future of the Global Regulatory Affairs Outsourcing Market .
Are you ready to discover more about the Regulatory Affairs Outsourcing Market?
The report provides an in-depth analysis of the leading companies operating in the global Regulatory Affairs Outsourcing Market. It includes a comparative assessment based on their product portfolios, business overviews, geographical footprint, strategic initiatives, market segment share, and SWOT analysis. Each company is profiled using a standardized format that includes:
Company Profiles
1.IQVIA
- Business Overview
- Company Snapshot
- Products Overview
- Company Market Share Analysis
- Company Coverage Portfolio
- Financial Analysis
- Recent Developments
- Merger and Acquisitions
- SWOT Analysis
2.ICON plc
3.Thermo Fisher Scientific (PPD)
4.Labcorp Drug Development
5.Syneos Health
6.Parexel International
7.Charles River Laboratories
8.Freyr Solutions
9.ProPharma Group
10.Others.
Conclusion
The global regulatory affairs outsourcing market size is estimated to grow from USD 6.9 billion in 2024 to USD 17.84 billion in 2035, at a compound annual growth rate (CAGR) of 9.02%, driven by increased regulatory complexities and a globalized clinical development landscape. Increasing regulatory complexities in the approval processes by regulatory bodies such as the U.S. Food and Drug Administration and European Medicines Agency are adding 15-20% to drug approval times, which is a key driver for regulatory affairs outsourcing. The increase in biologics and personalized medicine is another driver for regulatory affairs outsourcing, as these are complex areas requiring specialized regulatory affairs expertise. The Asia Pacific region is expected to see high growth in regulatory affairs outsourcing, driven by a 25% increase in clinical trial activities and a cost-effective outsourcing environment. Major regulatory affairs outsourcing companies such as IQVIA and ICON plc are investing in artificial intelligence-based automation and strategic partnerships to enhance their capabilities in regulatory affairs outsourcing services, which is a key trend in the regulatory affairs outsourcing market, with a shift towards integrated and technology-based outsourcing models, which are improving accuracy in regulatory affairs, reducing costs by 30%, and accelerating time-to-market for innovative therapies.
Other Industry Reports :
Top 25 Companies in Anti Icing Coating Market
Top 25 Companies in Antihistamine Drugs Market
Top 30 Companies in HPL Fire-Resistant Board Market
Top 30 Companies in Interventional Pulmonology Market
Top 25 Companies in Handheld Marijuana Vaporizer Market
Top 25 Companies in Hydroxychloroquine Market
About the Spherical Insights & Consulting
Spherical Insights & Consulting is a market research and consulting firm which provides actionable market research study, quantitative forecasting and trends analysis provides forward-looking insight especially designed for decision makers and aids ROI.
Which is catering to different industry such as financial sectors, industrial sectors, government organizations, universities, non-profits and corporations. The company's mission is to work with businesses to achieve business objectives and maintain strategic improvements.
CONTACT US:
For More Information on Your Target Market, Please Contact Us Below:
Phone: +1 303 800 4326 (the U.S.)
Phone: +91 90289 24100 (APAC)
Email: inquiry@sphericalinsights.com, sales@sphericalinsights.com
Contact Us: https://www.sphericalinsights.com/contact-us
Need help to buy this report?