Top 15 Companies in Global Biopharma Buffer Market (2025–2035): Competitive Analysis & Forecast
RELEASE DATE: Mar 2026 Author: Spherical InsightsRequest Free Sample Speak to Analyst
Introduction
The global biopharma buffer market refers to a critical backbone for the life sciences industry, providing specialized chemical solutions designed to maintain precise pH stability and molecular integrity during drug development. Defined by their ability to resist acidity or alkalinity changes, these buffers are indispensable in both upstream cell culture and downstream purification processes. The market is characterized by a significant shift toward high-purity, GMP-grade formulations that ensure the safety and efficacy of complex biologics. Recent innovations are redefining the landscape, particularly through the rise of ready-to-use (RTU) liquid buffers and single-use technologies. Manufacturers are increasingly moving away from traditional, labor-intensive manual mixing toward automated buffer preparation systems and modular, disposable delivery units. Furthermore, the integration of AI-driven formulation tools now allows researchers to predict optimal buffer stability and customize recipes for specific therapeutic profiles, such as mRNA or CAR-T cell therapies. The market is primarily fueled by the surging global demand for monoclonal antibodies, vaccines, and biosimilars. As chronic diseases become more prevalent, the expansion of biomanufacturing facilities especially in emerging hubs across the Asia-Pacific region acts as a powerful catalyst. Additionally, the transition toward personalized medicine requires highly specific, small-batch buffer solutions, further pushing the industry toward a more agile and technologically advanced ecosystem. This combination of rising R&D investment and a focus on operational efficiency positions the sector for robust, long-term growth.
Navigate Future Markets with Confidence: Insights from Spherical Insights LLP
The insights presented in this blog are derived from comprehensive market research conducted by Spherical Insights LLP, a trusted advisory partner to leading global enterprises. Backed by in-depth data analysis, expert forecasting, and industry-specific intelligence, our reports empower decision-makers to identify strategic growth opportunities in fast-evolving sectors. Clients seeking detailed market segmentation, competitive landscapes, regional outlooks, and future investment trends will find immense value in the full report. By leveraging our research, businesses can make informed decisions, gain a competitive edge, and stay ahead in the transition toward sustainable and profitable solutions.
Unlock exclusive market insights—Download the Brochure now and dive deeper into the future of the Biopharma Buffer Market.
Biopharma Buffer Market Size & Statistics
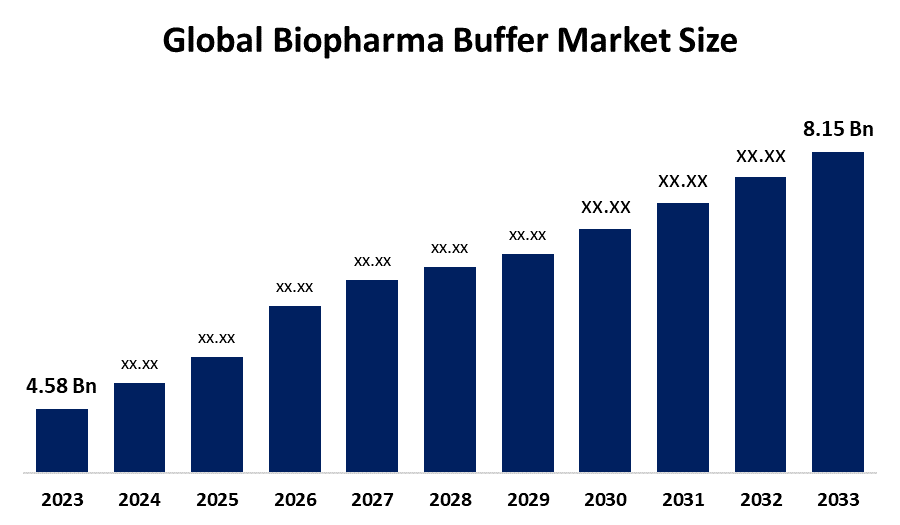
- The Market Size for Biopharma Buffer Was Estimated to be worth USD 4.58 Billion in 2024.
- The Market is Going to Expand at a CAGR of 7.6% between 2025 and 2035.
- The Global Biopharma Buffer Market Size is anticipated to reach USD 8.15 Billion by 2035.
- North America is expected to generate the highest demand during the forecast period in the Biopharma Buffer Market
- Asia Pacific is expected to grow the fastest during the forecast period in the Biopharma Buffer Market.
Regional Growth and Demand
Asia Pacific is expected to grow the fastest during the forecast period in the Biopharma Buffer market. Asia Pacific is expected to grow fastest due to massive infrastructure investments in China, India, and South Korea, which have become global hubs for biosimilar production and contract manufacturing. Additionally, favorable government initiatives and a shift toward localized drug development are accelerating the demand for high-purity buffering agents in this territory.
North America is expected to generate the highest demand during the forecast period in the Biopharma Buffer market. North America is expected to generate the highest demand due to its leadership, which is anchored by a dense concentration of major biopharmaceutical giants and world-class research institutions that command high volumes of specialized buffers. Furthermore, the region’s early adoption of advanced single-use technologies and a robust pipeline for cell and gene therapies continue to solidify its position as the primary revenue generator.
Top 10 Trends in the Biopharma Buffer Market
- Automation & AI in Buffer Preparation
- Transition Towards Continuous Bioprocessing
- Localization of Supply Chains & CDMO Partnerships
- Green Chemistry Approaches
- Advancements in Dry Powder Hydration
- Complete Eradication of Animal-Derived Components
- Smart Packaging and IoT- Enabled Logistics Tracking
- Stringent Multi- Compendial Regulatory Compliance
- Shift Toward Single-Use Technologies
- Adoption of In-Line Buffer Dilution and Conditioning
1.Automation & AI in Buffer Preparation
The integration of Artificial Intelligence (AI) and automated scheduling software is revolutionizing how buffers are prepared and managed on the factory floor. Advanced process simulators and finite capacity scheduling tools are now being deployed to predict exact buffer demands based on bioreactor output and downstream processing speeds. AI algorithms optimize the blending of complex buffer recipes by analyzing real-time sensor data, automatically adjusting flow rates to maintain precise ionic strength and pH parameters. This eliminates manual interventions, minimizes the risk of out-of-specification deviations, and ensures that buffer supply seamlessly matches the continuous rhythm of the broader manufacturing workflow.
2.Localization of Supply Chains & CDMO Partnerships
Recent global disruptions have exposed the vulnerabilities of heavily centralized, international supply chains. In response, biopharma companies are actively localizing their procurement of critical buffer salts and chemicals to ensure supply security. Alongside this geographical shift, there is a deepening reliance on Contract Development and Manufacturing Organizations (CDMOs). These specialized partners not only provide localized, agile buffer manufacturing but also offer the expertise needed to rapidly scale up production.
3.Green Chemistry Approaches
Driven by strict corporate Environmental, Social, and Governance (ESG) mandates, the buffer market is seeing a distinct shift toward green chemistry. Manufacturers are moving beyond simply reducing water usage to actively rethinking the chemical composition of the buffers themselves. This involves the development of biodegradable formulations and the elimination of hazardous or environmentally persistent stabilizing agents.
4.Complete Eradication of Animal-Derived Components
The demand for absolute safety and purity in biologic manufacturing has solidified the transition toward chemically defined, entirely animal-free buffer formulations. Historically, certain stabilizing agents or complex media components may have carried trace animal derivatives, which introduce the severe risk of adventitious viral contamination or prion transmission. Regulatory bodies now heavily favor, and often mandate, animal-origin-free (AOF) components for modern therapeutics.
5.Smart Packaging and IoT-Enabled Logistics Tracking
The logistics of transporting highly sensitive, GMP-grade liquid buffers have evolved dramatically with the integration of the Internet of Things (IoT) and smart packaging. These advanced shipping containers are equipped with digital sensors that continuously monitor critical environmental variables such as temperature fluctuations, humidity, and physical shock during transit. This real-time data is transmitted to cloud platforms, allowing manufacturers to verify that the buffer maintained its chemical integrity from the supplier’s warehouse to the bioreactor floor. This rigorous, tech-enabled chain of custody prevents the costly use of degraded buffers and satisfies the highest standards of regulatory quality assurance.
Empower your strategic planning:
Stay informed with the latest industry insights and market trends to identify new opportunities and drive growth in the biopharma buffer market. To explore more in-depth trends, insights, and forecasts, please refer to our detailed report.
Top 15 Companies Leading the Biopharma Buffer Market
- Thermo Fischer Scientific
- Merck KGaA
- Avantor
- Lonza Group
- Sartorius
- Cytiva
- Medix Biochemica
- Canvax Biotech
- Santa Cruz Biotechnology
- Masterflex Bioprocessing
- Prepared Biologics
- Molekula
- Taiwan Hopax Chemicals
- Sepragen Corporation
- Charles River Laboratories
1.Thermo Fischer Scientific
Headquarters: Waltham, Massachusetts, USA
Thermo Fischer Scientic, active in more than 50 countries, is deeply integrated into both the upstream and downstream bioprocessing workflows of therapeutic development. It is widely recognized as a principal supplier of critical buffering agents, leveraging its renowned Gibco brand portfolio. The company provides a comprehensive array of buffer formats tailored to the exact needs of biologic manufacturers. This includes everything from bulk, raw buffer salts to highly sophisticated, ready-to-use liquid buffer formulations that plug directly into modern single-use manufacturing lines.
2.Merck kGaA
Headquarters: Darmstadt, Germany
Merck kGaA, active in over 66 countries, is a comprehensive solutions provider, heavily influencing how the industry approaches pH regulation and molecule stability. The company's life science division offers an incredibly broad portfolio of high-purity biological buffers, primarily marketed under its SAFC and Emprove product lines. The Emprove program, in particular, provides manufacturers with extensive regulatory documentation for buffer salts, vastly simplifying the compliance process for complex biological therapies. Their buffer offerings span a wide range of chemistries, including TRIS, HEPES, and phosphate-based solutions, available in both highly refined dry powder formats and ready-to-use liquid configurations.
3.Avantor
Headquarters: Loveland, Colorado, USA
Avantor, active in over 30 countries, is a widely regarded as a cornerstone supplier of critical chemical solutions. The company is celebrated for its highly respected J.T.Baker brand, which represents a gold standard in high-purity buffer salts and biological reagents. Avantor’s primary activity in this sector revolves around the cGMP-compliant manufacturing and distribution of specialized buffering agents that are essential for both upstream cell culture environments and rigorous downstream purification processes. Their product portfolio is engineered to mitigate the risks of trace metal contamination, which can severely compromise the stability and yield of sensitive biologic drugs. In addition to supplying traditional dry powder buffer salts, Avantor has significantly expanded its capabilities in the realm of fluid handling and customized buffer preparation.
4.Cytiva
Headquarters: Marlborough, Massachusetts, United States
Cytiva, active in more than 40 countries, is profound and deeply integrated into the fundamental workflows of therapeutic production. The company is highly active in formulating, manufacturing, and supplying critical biological buffers, most notably through its widely respected HyClone brand portfolio. They offer an extensive array of high-quality buffering products, ranging from meticulously blended dry powder formats to ultra-pure, ready-to-use liquid formulations that are essential for maintaining molecular stability during downstream chromatography and ultrafiltration processes. Beyond providing the consumable chemical formulations, Cytiva is a pioneer in advanced buffer management technologies.
5.Taiwan Hopax Chemicals
Headquarters: Kaohsiung, Taiwan
Taiwan Hopax Chemicals, active in over 50 countries, is a premier manufacturer of Good's buffers. Formulated initially by Dr. Norman Good, these specific zwitterionic buffering agents such as HEPES, TRIS, PIPES, and MES are fundamentally essential to modern biological research and biomanufacturing. HOPAX actively focuses on the large-scale synthesis of these critical molecules, ensuring they meet the exceptionally stringent purity standards required for mammalian cell culture environments and complex downstream protein purification workflows.
Are you ready to discover more about the Biopharma Buffer market?
The report provides an in-depth analysis of the leading companies operating in the global Biopharma Buffer market. It includes a comparative assessment based on their product portfolios, business overviews, geographical footprint, strategic initiatives, market segment share, and SWOT analysis. Each company is profiled using a standardized format that includes:
Company Profiles
- Thermo Fischer Scientific
- Business Overview
- Company Snapshot
- Products Overview
- Company Market Share Analysis
- Company Coverage Portfolio
- Financial Analysis
- Recent Developments
- Merger and Acquisitions
- SWOT Analysis
- Merck KGaA
- Avantor
- Lonza Group
- Sartorius
- Cytiva
- Medix Biochemica
- Canvax Biotech
- Santa Cruz Biotechnology
- Others.
Conclusion
The global biopharma buffer market is witnessing robust growth, driven by the surging demand for complex biologics, rapid advancements in continuous biomanufacturing processes, and expanding investments in targeted personalized therapies. Biopharma buffers provide critical, high-purity chemical solutions for both upstream cell culture and rigorous downstream purification applications, ensuring molecular stability, precise pH control, and the ultimate efficacy of life-saving drugs. Innovations in ready-to-use (RTU) liquid formulations, single-use technologies (SUT), and intelligent in-line dilution systems further enhance operational efficiency and significantly reduce contamination risks. Leading companies such as Thermo Fisher Scientific, Merck KGaA, Avantor, Lonza Group, and Sartorius are expanding their global manufacturing footprints and technical capabilities, directly contributing to continuous market development. Overall, biopharma buffers play an absolutely vital role in advancing the safe, scalable, and highly efficient commercial production of modern biologic medicines globally.
About the Spherical Insights & Consulting
Spherical Insights & Consulting is a market research and consulting firm which provides actionable market research study, quantitative forecasting and trends analysis provides forward-looking insight especially designed for decision makers and aids ROI.
Which is catering to different industry such as financial sectors, industrial sectors, government organizations, universities, non-profits and corporations. The company's mission is to work with businesses to achieve business objectives and maintain strategic improvements.
CONTACT US:
For More Information on Your Target Market, Please Contact Us Below:
Phone: +1 303 800 4326 (the U.S.)
Phone: +91 90289 24100 (APAC)
Email: inquiry@sphericalinsights.com, sales@sphericalinsights.com
Contact Us: https://www.sphericalinsights.com/contact-us
Need help to buy this report?